Dual‑regulated oncolytic adenovirus carrying ERCC1‑siRNA gene possesses potent antitumor effect on ovarian cancer cells
- PMID: 38757346
- PMCID: PMC11129538
- DOI: 10.3892/mmr.2024.13245
Dual‑regulated oncolytic adenovirus carrying ERCC1‑siRNA gene possesses potent antitumor effect on ovarian cancer cells
Abstract
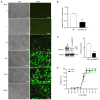
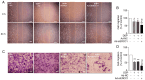
Ovarian cancer is a multifactorial and deadly disease. Despite significant advancements in ovarian cancer therapy, its incidence is on the rise and the molecular mechanisms underlying ovarian cancer invasiveness, metastasis and drug resistance remain largely elusive, resulting in poor prognosis. Oncolytic viruses armed with therapeutic transgenes of interest offer an attractive alternative to chemical drugs, which often face innate and acquired drug resistance. The present study constructed a novel oncolytic adenovirus carrying ERCC1 short interfering (si)RNA, regulated by hTERT and HIF promoters, termed Ad‑siERCC1. The findings demonstrated that this oncolytic adenovirus effectively inhibits the proliferation, migration and invasion of ovarian cancer cells. Furthermore, the downregulation of ERCC1 expression by siRNA ameliorates drug resistance to cisplatin (DDP) chemotherapy. It was found that Ad‑siERCC1 blocks the cell cycle in the G1 phase and enhances apoptosis through the PI3K/AKT‑caspase‑3 signaling pathways in SKOV3 cells. The results of the present study highlighted the critical effect of oncolytic virus Ad‑siERCC1 in inhibiting the survival of ovarian cancer cells and increasing chemotherapy sensitivity to DDP. These findings underscore the potent antitumor effect of Ad‑siERCC1 on ovarian cancers in vivo.
Keywords: ERCC1; adenovirus; hTERT/HIF; ovarian cancer; proliferation.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



Similar articles
-
Interfering with the expression of EEF1D gene enhances the sensitivity of ovarian cancer cells to cisplatin.BMC Cancer. 2022 Jun 8;22(1):628. doi: 10.1186/s12885-022-09699-7. BMC Cancer. 2022. PMID: 35672728 Free PMC article.
-
Lentivirus-Mediated RNAi silencing targeting ERCC1 reverses cisplatin resistance in cisplatin-resistant ovarian carcinoma cell line.DNA Cell Biol. 2015 Jul;34(7):497-502. doi: 10.1089/dna.2015.2805. Epub 2015 May 5. DNA Cell Biol. 2015. PMID: 25941922
-
A potent oncolytic adenovirus selectively blocks the STAT3 signaling pathway and potentiates cisplatin antitumor activity in ovarian cancer.Hum Gene Ther. 2012 Jan;23(1):32-45. doi: 10.1089/hum.2011.101. Epub 2011 Oct 14. Hum Gene Ther. 2012. PMID: 21875334
-
[ERCC1 as a Marker of Ovarian Cancer Resistance to Platinum Drugs].Antibiot Khimioter. 2015;60(3-4):42-50. Antibiot Khimioter. 2015. PMID: 26415382 Review. Russian.
-
Sindbis Virus Vaccine Platform: A Promising Oncolytic Virus-Mediated Approach for Ovarian Cancer Treatment.Int J Mol Sci. 2024 Mar 2;25(5):2925. doi: 10.3390/ijms25052925. Int J Mol Sci. 2024. PMID: 38474178 Free PMC article. Review.
Cited by
-
Oncolytic Viruses in Ovarian Cancer: Where Do We Stand? A Narrative Review.Pathogens. 2025 Feb 3;14(2):140. doi: 10.3390/pathogens14020140. Pathogens. 2025. PMID: 40005517 Free PMC article. Review.
-
The role of BUD31 in clear cell renal cell carcinoma: prognostic significance, alternative splicing, and tumor immune environment.Clin Exp Med. 2024 Aug 13;24(1):191. doi: 10.1007/s10238-024-01451-8. Clin Exp Med. 2024. PMID: 39136845 Free PMC article.
References
-
- Penny SM. Ovarian cancer: An overview. Radiol Technol. 2020;91:561–575. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

