Methodological Quality of Clinical Trials in Amyotrophic Lateral Sclerosis: A Systematic Review
- PMID: 38759021
- PMCID: PMC11307009
- DOI: 10.3233/JND-230217
Methodological Quality of Clinical Trials in Amyotrophic Lateral Sclerosis: A Systematic Review
Abstract
Background: More than 200 clinical trials have been performed worldwide in ALS so far, but no agents with substantial efficacy on disease progression have been found.
Objective: To describe the methodological quality of all clinical trials performed in ALS and published before December 31, 2022.
Methods: We conducted a systematic review following the Preferred Reporting Items for Systematic Reviews and Meta Analyses.
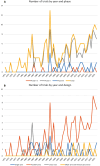
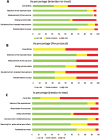
Results: 213 trials were included. 47.4% manuscripts described preclinical study evaluation, with a positive effect in all. 67.6% of trials were conducted with a parallel-arm design, while 12.7% were cross-over studies; 77% were randomized, while in 5.6% historical-controls were used for comparison. 70% of trials were double blind. Participant inclusion allowed forced vital capacity (or corresponding slow vital capacity)<50% in 15% cases, between 55-65% in 21.6%, between 70-80% in 14.1% reports, and 49.3% of the evaluated manuscripts did not provide a minimum value for respiratory capacity at inclusion. Disease duration was < 6-months in 6 studies, 7-36 months in 68, 37-60 months in 24, 8 trials requested more than 1-month of disease duration, while in 107 reports a disease duration was not described. Dropout rate was ≥20% in 30.5% trials, while it was not reported for 8.5%.
Conclusion: The methodological quality of the included studies was highly variable. Major issues to be addressed in future ALS clinical trials include: the requirement for standard animal toxicology and phase I studies, the resource-intensive nature of phase II-III studies, adequate study methodology and design, a good results reporting.
Keywords: Clinical trial; amyotrophic lateral sclerosis; drugs; methodology; systematic review.
Conflict of interest statement
No conflict of interest to be declared for the present review.
Figures
References
-
- Sackett D, Straus S, Richardson S, Rosenberg W, Haynes R. Evidence-based medicine: how to practice and teach EBM. 2000.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous