Tumor marker-based RecistTM is superior to RECIST as criteria to predict the long-term benefits of targeted therapy in advanced non-small-cell lung cancer with driver gene mutations
- PMID: 38761505
- PMCID: PMC11127532
- DOI: 10.1016/j.neo.2024.101006
Tumor marker-based RecistTM is superior to RECIST as criteria to predict the long-term benefits of targeted therapy in advanced non-small-cell lung cancer with driver gene mutations
Abstract
Background: Tyrosine kinase inhibitors (TKIs) are standard first-line treatments for advanced non-small-cell lung cancer (NSCLC) with driver gene mutations. The Response Evaluation Criteria in Solid Tumors (RECIST) are limited in predicting long-term patient benefits. A tumour marker-based evaluation criteria, RecistTM, was used to investigate the potential for assessing targeted-therapy efficacy in lung cancer treatment.
Methods: We retrospectively analysed patients with stage IIIA-IV NSCLC and driver gene mutations, whose baseline tumour marker levels exceeded the pre-treatment cut-off value three-fold and who received TKI-targeted therapy as a first-line treatment. We compared efficacy, progression-free survival (PFS), and overall survival (OS) between RecistTM and RECIST.
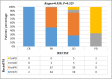
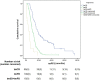
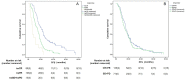
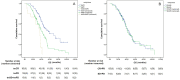
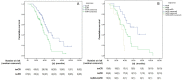
Findings: The median PFS and OS differed significantly among treatment-response subgroups based on RecistTM but not RECIST. The predicted 1-, 2-, and 3-year disease-progression risk, according to area under the receiver operating characteristic curve, as well as the 1-, 3-, and 5-year mortality risk, differed significantly between RecistTM and RECIST. The median PFS and OS of tmCR according to RecistTM, was significantly longer than (CR+PR) according to RECIST. Imaging analysis revealed that the ΔPFS was 11.27 and 6.17 months in the intervention and non-intervention groups, respectively, suggesting that earlier intervention could extend patients' PFS.
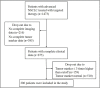
Interpretation: RecistTM can assess targeted-therapy efficacy in patients with advanced NSCLC and driver gene mutations, along with tumour marker abnormalities. RecistTM surpasses RECIST in predicting short- and long-term patient benefits, and allows the early identification of patients resistant to targeted drugs, enabling prompt intervention and extending the imaging-demonstrated time to progression.
Keywords: Non-small-cell lung cancer; Overall survival; Progression-free survival; RECIST; RecistTM.
Copyright © 2024. Published by Elsevier Inc.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


Similar articles
-
Elevated tumor markers for monitoring tumor response to immunotherapy.EClinicalMedicine. 2022 Apr 8;46:101381. doi: 10.1016/j.eclinm.2022.101381. eCollection 2022 Apr. EClinicalMedicine. 2022. PMID: 35434583 Free PMC article.
-
RECIST progression patterns during EGFR tyrosine kinase inhibitor treatment of advanced non-small cell lung cancer patients harboring an EGFR mutation.Lung Cancer. 2015 Dec;90(3):477-83. doi: 10.1016/j.lungcan.2015.09.025. Epub 2015 Oct 9. Lung Cancer. 2015. PMID: 26604031
-
Optimal tumor shrinkage predicts long-term outcome in advanced nonsmall cell lung cancer (NSCLC) treated with target therapy: Result from 3 clinical trials of advanced NSCLC by 1 institution.Medicine (Baltimore). 2016 Aug;95(31):e4176. doi: 10.1097/MD.0000000000004176. Medicine (Baltimore). 2016. PMID: 27495021 Free PMC article.
-
Effect of smoking status on progression-free and overall survival in non-small cell lung cancer patients receiving erlotinib or gefitinib: a meta-analysis.J Clin Pharm Ther. 2015 Dec;40(6):661-71. doi: 10.1111/jcpt.12332. Epub 2015 Nov 17. J Clin Pharm Ther. 2015. PMID: 26573867
-
Mixed responses to first-line alectinib in non-small cell lung cancer patients with rare ALK gene fusions: A case series and literature review.J Cell Mol Med. 2021 Oct;25(19):9476-9481. doi: 10.1111/jcmm.16897. Epub 2021 Sep 19. J Cell Mol Med. 2021. PMID: 34541785 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

