Immune cell profiling in intestinal transplantation
- PMID: 38762429
- PMCID: PMC11283363
- DOI: 10.1016/j.humimm.2024.110808
Immune cell profiling in intestinal transplantation
Abstract
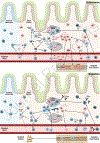
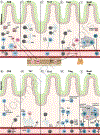
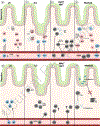
Since the first published case study of human intestinal transplantation in 1967, there have been significant studies of intestinal transplant immunology in both animal models and humans. An improved understanding of the profiles of different immune cell subsets is critical for understanding their contributions to graft outcomes. While different studies have focused on the contribution of one or a few subsets to intestinal transplant, no study has integrated these data for a comprehensive overview of immune dynamics after intestinal transplant. Here, we provide a systematic review of the literature on different immune subsets and discuss their roles in intestinal transplant outcomes on multiple levels, focusing on chimerism and graft immune reconstitution, clonal alloreactivity, and cell phenotype. In Sections 1, 2 and 3, we lay out a shared framework for understanding intestinal transplant, focusing on the mechanisms of rejection or tolerance in the context of mucosal immunology and illustrate the unique role of the bidirectional graft-versus-host (GvH) and host-versus-graft (HvG) alloresponse. In Sections 4, 5 and 6, we further expand upon these concepts as we discuss the contribution of different cell subsets to intestinal transplant. An improved understanding of intestinal transplantation immunology will bring us closer to maximizing the potential of this important treatment.
Keywords: Bidirectional alloresponse; Chimerism; Immune cell profiling; Intestinal transplantation.
Copyright © 2024 American Society for Histocompatibility and Immunogenetics. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest J.F. served as a Scientific Consultant for Adaptive Biotechnologies Corp. from June 2022 to May 2023. J.F. is a co-investigator for a research grant provided by Ossium Heath Inc. and a co-inventor for a patent issued to Ossium Heath Inc. and the Trustees of Columbia University in the City of New York.
Figures
Similar articles
-
Hematopoietic cell-based and non-hematopoietic cell-based strategies for immune tolerance induction in living-donor renal transplantation: A systematic review.Transplant Rev (Orlando). 2023 Dec;37(4):100792. doi: 10.1016/j.trre.2023.100792. Epub 2023 Aug 19. Transplant Rev (Orlando). 2023. PMID: 37709652
-
Mucosal Mixed Lymphocyte Reaction Assay Using Intestinal Lymphocytes as a Biomarker for Intestinal Transplant Tolerance Development.Transplantation. 2025 Jul 1;109(7):1175-1183. doi: 10.1097/TP.0000000000005348. Epub 2025 Feb 19. Transplantation. 2025. PMID: 39966995
-
Bone marrow versus peripheral blood allogeneic haematopoietic stem cell transplantation for haematological malignancies in adults.Cochrane Database Syst Rev. 2024 Nov 7;11(11):CD010189. doi: 10.1002/14651858.CD010189.pub3. Cochrane Database Syst Rev. 2024. PMID: 39508306
-
Interventions for increasing immunosuppressant medication adherence in solid organ transplant recipients.Cochrane Database Syst Rev. 2022 Sep 12;9(9):CD012854. doi: 10.1002/14651858.CD012854.pub2. Cochrane Database Syst Rev. 2022. PMID: 36094829 Free PMC article.
-
Polyclonal and monoclonal antibodies for treating acute rejection episodes in kidney transplant recipients.Cochrane Database Syst Rev. 2017 Jul 20;7(7):CD004756. doi: 10.1002/14651858.CD004756.pub4. Cochrane Database Syst Rev. 2017. PMID: 28731207 Free PMC article.
References
-
- Kataoka K: The intestinal microbiota and its role in human health and disease. J Med Invest 2016;63:27. - PubMed
-
- DiBaise J: Management of short bowel syndrome in adults. UpToDate 2023.
-
- Iyer KR, Winkler M, Zubizarreta N, Nisenholtz M, Lucero K, Lubarda J: Knowledge of chronic intestinal failure among US gastroenterologists: Cause for concern and learning opportunity. JPEN J Parenter Enteral Nutr 2022;46:730. - PubMed
-
- Khan FA, Selvaggi G: Overview of intestinal and multivisceral transplantation. UpToDate 2023.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

