Comparative microbiome analysis in cystic fibrosis and non-cystic fibrosis bronchiectasis
- PMID: 38762736
- PMCID: PMC11102160
- DOI: 10.1186/s12931-024-02835-w
Comparative microbiome analysis in cystic fibrosis and non-cystic fibrosis bronchiectasis
Abstract
Background: Bronchiectasis is a condition characterized by abnormal and irreversible bronchial dilation resulting from lung tissue damage and can be categorized into two main groups: cystic fibrosis (CF) and non-CF bronchiectasis (NCFB). Both diseases are marked by recurrent infections, inflammatory exacerbations, and lung damage. Given that infections are the primary drivers of disease progression, characterization of the respiratory microbiome can shed light on compositional alterations and susceptibility to antimicrobial drugs in these cases compared to healthy individuals.
Methods: To assess the microbiota in the two studied diseases, 35 subjects were recruited, comprising 10 NCFB and 13 CF patients and 12 healthy individuals. Nasopharyngeal swabs and induced sputum were collected, and total DNA was extracted. The DNA was then sequenced by the shotgun method and evaluated using the SqueezeMeta pipeline and R.
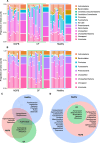
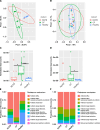
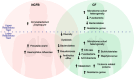
Results: We observed reduced species diversity in both disease cohorts, along with distinct microbial compositions and profiles of antimicrobial resistance genes, compared to healthy individuals. The nasopharynx exhibited a consistent microbiota composition across all cohorts. Enrichment of members of the Burkholderiaceae family and an increased Firmicutes/Bacteroidetes ratio in the CF cohort emerged as key distinguishing factors compared to NCFB group. Staphylococcus aureus and Prevotella shahii also presented differential abundance in the CF and NCFB cohorts, respectively, in the lower respiratory tract. Considering antimicrobial resistance, a high number of genes related to antibiotic efflux were detected in both disease groups, which correlated with the patient's clinical data.
Conclusions: Bronchiectasis is associated with reduced microbial diversity and a shift in microbial and resistome composition compared to healthy subjects. Despite some similarities, CF and NCFB present significant differences in microbiome composition and antimicrobial resistance profiles, suggesting the need for customized management strategies for each disease.
Keywords: Bronchiectasis; Cystic fibrosis; Microbiome; Non-cystic fibrosis; Resistome.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
-
- Aliberti S, Goeminne PC, O’Donnell AE, Aksamit TR, Al-Jahdali H, Barker AF, et al. Criteria and definitions for the radiological and clinical diagnosis of bronchiectasis in adults for use in clinical trials: international consensus recommendations. Lancet Respir Med. 2022;10:298–306. doi: 10.1016/S2213-2600(21)00277-0. - DOI - PubMed
Publication types
MeSH terms
Grants and funding
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 21/2551-0000103-8/Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
- 2021-0322/Financiamento e Incentivo à Pesquisa - Hospital de Clínicas de Porto Alegre - Fipe/HCPA
LinkOut - more resources
Full Text Sources
Medical

