Adding 6 months of androgen deprivation therapy to postoperative radiotherapy for prostate cancer: a comparison of short-course versus no androgen deprivation therapy in the RADICALS-HD randomised controlled trial
- PMID: 38763154
- PMCID: PMC7616360
- DOI: 10.1016/S0140-6736(24)00548-8
Adding 6 months of androgen deprivation therapy to postoperative radiotherapy for prostate cancer: a comparison of short-course versus no androgen deprivation therapy in the RADICALS-HD randomised controlled trial
Abstract
Background: Previous evidence indicates that adjuvant, short-course androgen deprivation therapy (ADT) improves metastasis-free survival when given with primary radiotherapy for intermediate-risk and high-risk localised prostate cancer. However, the value of ADT with postoperative radiotherapy after radical prostatectomy is unclear.
Methods: RADICALS-HD was an international randomised controlled trial to test the efficacy of ADT used in combination with postoperative radiotherapy for prostate cancer. Key eligibility criteria were indication for radiotherapy after radical prostatectomy for prostate cancer, prostate-specific antigen less than 5 ng/mL, absence of metastatic disease, and written consent. Participants were randomly assigned (1:1) to radiotherapy alone (no ADT) or radiotherapy with 6 months of ADT (short-course ADT), using monthly subcutaneous gonadotropin-releasing hormone analogue injections, daily oral bicalutamide monotherapy 150 mg, or monthly subcutaneous degarelix. Randomisation was done centrally through minimisation with a random element, stratified by Gleason score, positive margins, radiotherapy timing, planned radiotherapy schedule, and planned type of ADT, in a computerised system. The allocated treatment was not masked. The primary outcome measure was metastasis-free survival, defined as distant metastasis arising from prostate cancer or death from any cause. Standard survival analysis methods were used, accounting for randomisation stratification factors. The trial had 80% power with two-sided α of 5% to detect an absolute increase in 10-year metastasis-free survival from 80% to 86% (hazard ratio [HR] 0·67). Analyses followed the intention-to-treat principle. The trial is registered with the ISRCTN registry, ISRCTN40814031, and ClinicalTrials.gov, NCT00541047.
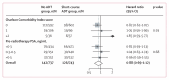
Findings: Between Nov 22, 2007, and June 29, 2015, 1480 patients (median age 66 years [IQR 61-69]) were randomly assigned to receive no ADT (n=737) or short-course ADT (n=743) in addition to postoperative radiotherapy at 121 centres in Canada, Denmark, Ireland, and the UK. With a median follow-up of 9·0 years (IQR 7·1-10·1), metastasis-free survival events were reported for 268 participants (142 in the no ADT group and 126 in the short-course ADT group; HR 0·886 [95% CI 0·688-1·140], p=0·35). 10-year metastasis-free survival was 79·2% (95% CI 75·4-82·5) in the no ADT group and 80·4% (76·6-83·6) in the short-course ADT group. Toxicity of grade 3 or higher was reported for 121 (17%) of 737 participants in the no ADT group and 100 (14%) of 743 in the short-course ADT group (p=0·15), with no treatment-related deaths.
Interpretation: Metastatic disease is uncommon following postoperative bed radiotherapy after radical prostatectomy. Adding 6 months of ADT to this radiotherapy did not improve metastasis-free survival compared with no ADT. These findings do not support the use of short-course ADT with postoperative radiotherapy in this patient population.
Funding: Cancer Research UK, UK Research and Innovation (formerly Medical Research Council), and Canadian Cancer Society.
Copyright © 2024 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests AN reports honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Merck. ADC reports research grants for the STAMPEDE trial from Janssen, Astellas, Novartis, Sanofi, and Clovis. ACT reports research grants to their institution from Elekta, Varian, and Accuracy; honoraria for talks from Elekta, Accuracy, and Janssen; travel assistance for radiotherapy conferences from Elekta; serving on the data safety board for two academic trials; and roles as the Chair of the MR-Linac Consortium and the genitourinary lead editor for the International Journal of Radiation Oncology, Biology, Physics. AC reports receiving grants or contracts from the National Institute for Health and Care Research, Manchester Biomedical Research Centre, Cancer Research UK, Medical Research Council (MRC) UK, Prostate Cancer UK, Bayer UK, and Elekta; and payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from the American Society for Radiation Oncology, the American Society of Clinical Oncology, Cancer Research UK, Roche, AstraZeneca, and Bristol Myers Squibb (BMS). CNC reports support for the present manuscript from the Canadian Cancer Trials Group; and payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Bayer Corp, Knight Therapeutics, and AbbVie. CLG reports participating in the independent data monitoring and steering committee for the BARCODE study at the Institute of Cancer Research in London (principal investigator Prof Ros Eeles). CCP reports consulting fees from AAA, ITM Radiopharma, Myovant, and Clarity Pharmaceuticals to his institution; and payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Janssen and Bayer to his institution. CM reports that they are employed by University College London (UCL) as Trial Manager for RADICALS. FS reports grants or contracts from Janssen, Bayer, Merck, Pfizer, Astellas, BMS, Novartis, Sanofi, and AstraZeneca to his institution; consulting fees from Janssen, Bayer, Astellas, BMS, Novartis, Sanofi, AstraZeneca, Merck, and Pfizer to him; and payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Janssen, Bayer, Myovant, Astellas, BMS, Novartis, Sanofi, AstraZeneca, Merck, and Pfizer to him. LCB reports their previous Institute of Cancer role was funded in part by a Biomedical Research Centre grant which was paid to the institution; the role was not connected to the RADICALS trial. MRS reports research grants and biomarker testing costs, all to their institution and all active in the past 36 months but on research outside of this research, from Astellas, Clovis Oncology, Janssen, Novartis, and Sanofi-Aventis; consulting fees from Eli Lilly; speaker fees at a clinical trial statistics training meeting for clinicians (no discussion of particular drugs) from Lilly Oncology, Janssen, and Eisai; and is an independent member of many independent data monitoring committees but all for academic sponsors and unpaid. NWC reports honoraria for lectures, advisory boards, and symposia from AstraZeneca, Janssen, Bayer, and Pfizer; support for travel to and attendance at a European meeting from Bayer; participation on the independent data monitoring committee for the Probio trial (Karolinska), and the trial steering committee for the Capi 28 trial (AstraZeneca) and the STAMPEDE trial (MRC); and is the Joint National Clinical Lead for the National Prostate Cancer Audit. PWMC reports grants or contracts from the Canadian Association of Radiation Oncology ACURA program and the Canadian Institute of Health Research; payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Pfizer and EMD Serono; and participation on a data safety monitoring board or advisory board from TerSera, AbbVie, Tolmar, and Knight Therapeutics. PMP reports participation on a data safety monitoring board or advisory board for AAA Nordic, MSD, and Pfizer Denmark. SCM reports honoraria from Janssen, Astellas, Knight Therapeutics, AMGEN Pharmaceutical, AstraZeneca, AbbVie, and Bayer; and support for attending meetings or travel from TerSera and Sanofi. VK reports a Cancer Research UK grant for the CORE trial to his institution; honoraria from Accuracy, Astellas, Bayer, and Boston Scientific; meeting attendance support from Accuray, Astellas, AstraZeneca, Bayer, BMS, Boston Scientific, Janssen, Merck Sharp & Dohme, and Norvatis; is an unpaid member of the independent data monitoring committee for the EPIC trial and the advisory board for Bayer and Janssen; and is the unpaid Chair of National Cancer Research Institute Penile subgroup of the Bladder and Renal Clinical Studies Group. WRC reports consulting fees from Bayer and Janssen; payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Astellas, Bayer, Janssen, AAA Novartis, and Myriad Genetics; and support for attending meetings or travel from Janssen, AAA Novartis, and Bayer. All other authors declare no competing interests.
Figures

Comment in
-
Androgen deprivation therapy combined with postoperative radiotherapy for prostate cancer management.Lancet. 2024 Jun 1;403(10442):2353-2355. doi: 10.1016/S0140-6736(24)00802-X. Epub 2024 May 16. Lancet. 2024. PMID: 38763152 No abstract available.
References
-
- Kishan AU, Sun Y, Hartman H, et al. Androgen deprivation therapy use and duration with definitive radiotherapy for localised prostate cancer: an individual patient data meta-analysis. Lancet Oncol. 2022;23:304–16. - PubMed
-
- Carrie C, Magné N, Burban-Provost P, et al. Short-term androgen deprivation therapy combined with radiotherapy as salvage treatment after radical prostatectomy for prostate cancer (GETUG-AFU 16): a 112-month follow-up of a phase 3, randomised trial. Lancet Oncol. 2019;20:1740–49. - PubMed
-
- Taylor LG, Canfield SE, Du XL. Review of major adverse effects of androgen-deprivation therapy in men with prostate cancer. Cancer. 2009;115:2388–99. - PubMed
-
- Lee LW, Clarke NW, Ramani VA, Cowan RA, Wylie JP, Logue JP. Adjuvant and salvage treatment after radical prostatectomy: current practice in the UK. Prostate Cancer Prostatic Dis. 2005;8:229–34. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

