Protective Role of the Podocyte IL-15 / STAT5 Pathway in Focal Segmental Glomerulosclerosis
- PMID: 38765560
- PMCID: PMC11101713
- DOI: 10.1016/j.ekir.2024.01.010
Protective Role of the Podocyte IL-15 / STAT5 Pathway in Focal Segmental Glomerulosclerosis
Abstract
Introduction: During glomerular diseases, podocyte-specific pathways can modulate the intensity of histological disease and prognosis. The therapeutic targeting of these pathways could thus improve the management and prognosis of kidney diseases. The Janus Kinase/Signal Transducer and Activator of Transcription (JAK/STAT) pathway, classically described in immune cells, has been recently described in detail in intrinsic kidney cells.
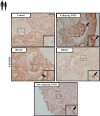
Methods: We describe STAT5 expression in human kidney biopsies from patients with focal segmental glomerulosclerosis (FSGS) and studied mice with a podocyte-specific Stat5 deletion in experimental glomerular diseases.
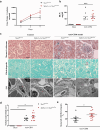
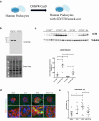
Results: Here, we show, for the first time, that STAT5 is activated in human podocytes in FSGS. In addition, podocyte-specific Stat5 inactivation aggravates the structural and functional alterations in a mouse model of FSGS. This could be due, at least in part, to an inhibition of autophagic flux. Finally, interleukin 15 (IL-15), a classical activator of STAT5 in immune cells, increases STAT5 phosphorylation in human podocytes, and its administration alleviates glomerular injury in vivo by maintaining autophagic flux in podocytes.
Conclusion: Activating podocyte STAT5 with commercially available IL-15 represents a potential new therapeutic avenue for FSGS.
Keywords: IL-15; autophagy; cell signaling; cytokines; focal segmental glomerulosclerosis; podocyte.
© 2024 International Society of Nephrology. Published by Elsevier Inc.
Figures



References
LinkOut - more resources
Full Text Sources
Miscellaneous

