Post-resolution macrophages shape long-term tissue immunity and integrity in a mouse model of pneumococcal pneumonia
- PMID: 38773113
- PMCID: PMC11109210
- DOI: 10.1038/s41467-024-48138-y
Post-resolution macrophages shape long-term tissue immunity and integrity in a mouse model of pneumococcal pneumonia
Abstract
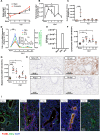
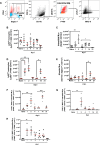
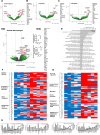
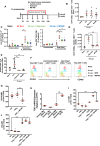
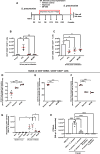
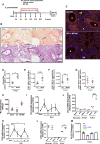
Resolving inflammation is thought to return the affected tissue back to homoeostasis but recent evidence supports a non-linear model of resolution involving a phase of prolonged immune activity. Here we show that within days following resolution of Streptococcus pneumoniae-triggered lung inflammation, there is an influx of antigen specific lymphocytes with a memory and tissue-resident phenotype as well as macrophages bearing alveolar or interstitial phenotype. The transcriptome of these macrophages shows enrichment of genes associated with prostaglandin biosynthesis and genes that drive T cell chemotaxis and differentiation. Therapeutic depletion of post-resolution macrophages, inhibition of prostaglandin E2 (PGE2) synthesis or treatment with an EP4 antagonist, MF498, reduce numbers of lung CD4+/CD44+/CD62L+ and CD4+/CD44+/CD62L-/CD27+ T cells as well as their expression of the α-integrin, CD103. The T cells fail to reappear and reactivate upon secondary challenge for up to six weeks following primary infection. Concomitantly, EP4 antagonism through MF498 causes accumulation of lung macrophages and marked tissue fibrosis. Our study thus shows that PGE2 signalling, predominantly via EP4, plays an important role during the second wave of immune activity following resolution of inflammation. This secondary immune activation drives local tissue-resident T cell development while limiting tissue injury.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

