Combination SGLT2 Inhibitor and Glucagon Receptor Antagonist Therapy in Type 1 Diabetes: A Randomized Clinical Trial
- PMID: 38776437
- PMCID: PMC11664189
- DOI: 10.2337/dc24-0212
Combination SGLT2 Inhibitor and Glucagon Receptor Antagonist Therapy in Type 1 Diabetes: A Randomized Clinical Trial
Abstract
Objective: To examine the effects of insulin-adjunctive therapy with a sodium-glucose cotransporter 2 (SGLT2) inhibitor and a glucagon receptor antagonist (GRA) on glycemia, insulin use, and ketogenesis during insulinopenia in type 1 diabetes.
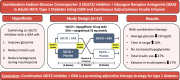
Research design and methods: In a randomized, double-blind, placebo-controlled, crossover trial we assessed the effects of adjunctive SGLT2 inhibitor therapy (dapagliflozin 10 mg daily) alone and in combination with the GRA volagidemab (70 mg weekly) in 12 adults with type 1 diabetes. Continuous glucose monitoring, insulin dosing, and insulin withdrawal tests (IWT) for measurement of glucose and ketogenesis during insulinopenia were completed during insulin-only (Baseline), SGLT2 inhibitor, and combination (SGLT2 inhibitor + GRA) therapy periods.
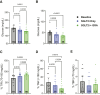
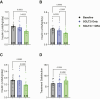
Results: Average glucose and percent time with glucose in range (70-180 mg/dL) improved with combination therapy versus Baseline and SGLT2 inhibitor (131 vs. 150 and 138 mg/dL [P < 0.001 and P = 0.01] and 86% vs. 70% and 78% [P < 0.001 and P = 0.03], respectively) without increased hypoglycemia. Total daily insulin use decreased with combination therapy versus Baseline and SGLT2 inhibitor (0.41 vs. 0.56 and 0.52 units/kg/day [P < 0.001 and P = 0.002]). Peak β-hydroxybutyrate levels during IWT were lower with combination therapy than with SGLT2 inhibitor (2.0 vs. 2.4 mmol/L; P = 0.048) and similar to levels reached during the Baseline testing period (2.1 mmol/L). Participants reported enhanced treatment acceptability and satisfaction with combination therapy.
Conclusions: Glucagon antagonism enhances the therapeutic effects of SGLT2 inhibition in type 1 diabetes. Combination therapy improves glycemic control, reduces insulin dosing, and suggests a strategy to unlock the benefits of SGLT2 inhibitors while mitigating the risk of diabetic ketoacidosis.
© 2024 by the American Diabetes Association.
Conflict of interest statement
Figures

References
-
- Pettus JH, Zhou FL, Shepherd L, et al. Incidences of severe hypoglycemia and diabetic ketoacidosis and prevalence of microvascular complications stratified by age and glycemic control in U.S. adult patients with type 1 diabetes: a real-world study. Diabetes Care 2019;42:2220–2227 - PubMed
-
- Goldenberg RM, Gilbert JD, Hramiak IM, Woo VC, Zinman B. Sodium-glucose co-transporter inhibitors, their role in type 1 diabetes treatment and a risk mitigation strategy for preventing diabetic ketoacidosis: The STOP DKA Protocol. Diabetes Obes Metab 2019;21:2192–2202 - PubMed
-
- Tucker ME. New York: Medscape. FDA turns down sotagliflozin for type 1 diabetes, 2019. Accessed 12 March 2020. Available from https://www.medscape.com/viewarticle/910851
Publication types
MeSH terms
Substances
Grants and funding
- Diabetes Research Connection
- T32 DK007044/DK/NIDDK NIH HHS/United States
- DK020593/Vanderbilt Diabetes Research and Training Center, Vanderbilt University Medical Center
- P30 DK063491/DK/NIDDK NIH HHS/United States
- K12 DK133995/DK/NIDDK NIH HHS/United States
- P30 DK020579/DK/NIDDK NIH HHS/United States
- K23 DK123392/DK/NIDDK NIH HHS/United States
- K12DK133995/National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health
- 2T32DK007044-41/NH/NIH HHS/United States
- UL1TR001442/UC San Diego Altman Clinical and Translational Research Institute
- UL1 TR001442/TR/NCATS NIH HHS/United States
- P30 DK020593/DK/NIDDK NIH HHS/United States
- 5-ECR-2020-950-A-N/JDRF/United States

