Surprising multifunctionality of a Tritonia swim CPG neuron: C2 drives the early phase of postswim crawling despite being silent during the behavior
- PMID: 38777746
- PMCID: PMC11381120
- DOI: 10.1152/jn.00001.2024
Surprising multifunctionality of a Tritonia swim CPG neuron: C2 drives the early phase of postswim crawling despite being silent during the behavior
Abstract
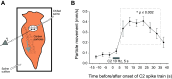
In response to a suitably aversive skin stimulus, the marine mollusk Tritonia diomedea launches an escape swim followed by several minutes of high-speed crawling. The two escape behaviors are highly dissimilar: whereas the swim is a muscular behavior involving alternating ventral and dorsal whole body flexions, the crawl is a nonrhythmic gliding behavior mediated by the beating of foot cilia. The serotonergic dorsal swim interneurons (DSIs) are members of the swim central pattern generator (CPG) and also strongly drive crawling. Although the swim network is very well understood, the Tritonia crawling network to date comprises only three neurons: the DSIs and pedal neurons 5 and 21 (Pd5 and Pd21). Since Tritonia's swim network has been suggested to have arisen from a preexisting crawling network, we examined the possible role that another swim CPG neuron, C2, may play in crawling. Because of its complete silence in the postswim crawling period, C2 had not previously been considered to play a role in driving crawling. However, semi-intact preparation experiments demonstrated that a brief C2 spike train surprisingly and strongly drives the foot cilia for ∼30 s, something that cannot be explained by its synaptic connections to Pd5 and Pd21. Voltage-sensitive dye (VSD) imaging in the pedal ganglion identified many candidate crawling motor neurons that fire at an elevated rate after the swim and also revealed several pedal neurons that are strongly excited by C2. It is intriguing that unlike the DSIs, which fire tonically after the swim to drive crawling, C2 does so despite its postswim silence.NEW & NOTEWORTHY Tritonia swim central pattern generator (CPG) neuron C2 surprisingly and strongly drives the early phase of postswim crawling despite being silent during this period. In decades of research, C2 had not been suspected of driving crawling because of its complete silence after the swim. Voltage-sensitive dye imaging revealed that the Tritonia crawling motor network may be much larger than previously known and also revealed that many candidate crawling neurons are excited by C2.
Keywords: invertebrate; multifunctional; neural network.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures





Comment in
-
Circuits and the single neuron.J Neurophysiol. 2024 Sep 1;132(3):765-766. doi: 10.1152/jn.00312.2024. Epub 2024 Jul 31. J Neurophysiol. 2024. PMID: 39081212 Free PMC article. No abstract available.
-
Multifunctionality of human linguistic neurons (circuits and single neurons).J Neurophysiol. 2025 Jan 1;133(1):1-2. doi: 10.1152/jn.00543.2024. Epub 2024 Nov 26. J Neurophysiol. 2025. PMID: 39589814 No abstract available.
References
-
- Frost WN, Hoppe TA, Wang J, Tian LM. Swim initiation neurons in Tritonia diomedea. Am Zool 41: 952–961, 2001. doi: 10.1093/icb/41.4.952. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

