Trends of use and characterisation of anti-dementia drugs users: a large multinational-network population-based study
- PMID: 38783756
- PMCID: PMC11116820
- DOI: 10.1093/ageing/afae106
Trends of use and characterisation of anti-dementia drugs users: a large multinational-network population-based study
Abstract
Background: An updated time-trend analysis of anti-dementia drugs (ADDs) is lacking. The aim of this study is to assess the incident rate (IR) of ADD in individuals with dementia using real-world data.
Setting: Primary care data (country/database) from the UK/CPRD-GOLD (2007-20), Spain/SIDIAP (2010-20) and the Netherlands/IPCI (2008-20), standardised to a common data model.
Methods: Cohort study. Participants: dementia patients ≥40 years old with ≥1 year of previous data. Follow-up: until the end of the study period, transfer out of the catchment area, death or incident prescription of rivastigmine, galantamine, donepezil or memantine. Other variables: age/sex, type of dementia, comorbidities. Statistics: overall and yearly age/sex IR, with 95% confidence interval, per 100,000 person-years (IR per 105 PY (95%CI)).
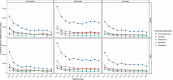
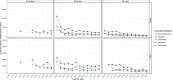
Results: We identified a total of (incident anti-dementia users/dementia patients) 41,024/110,642 in UK/CPRD-GOLD, 51,667/134,927 in Spain/SIDIAP and 2,088/17,559 in the Netherlands/IPCI.In the UK, IR (per 105 PY (95%CI)) of ADD decreased from 2007 (30,829 (28,891-32,862)) to 2010 (17,793 (17,083-18,524)), then increased up to 2019 (31,601 (30,483 to 32,749)) and decrease in 2020 (24,067 (23,021-25,148)). In Spain, IR (per 105 PY (95%CI)) of ADD decreased by 72% from 2010 (51,003 (49,199-52,855)) to 2020 (14,571 (14,109-15,043)). In the Netherlands, IR (per 105 PY (95%CI)) of ADD decreased by 77% from 2009 (21,151 (14,967-29,031)) to 2020 (4763 (4176-5409)). Subjects aged ≥65-79 years and men (in the UK and the Netherlands) initiated more frequently an ADD.
Conclusions: Treatment of dementia remains highly heterogeneous. Further consensus in the pharmacological management of patients living with dementia is urgently needed.
Keywords: cohort studies; dementia; drug utilization studies; older people.
© The Author(s) 2024. Published by Oxford University Press on behalf of the British Geriatrics Society. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Conflict of interest statement
KV and MM workfor a research group who, in the past 3 years, received unconditional research grants from Chiesi, UCB, Amgen, J&J and the EMA, none of which relate to the content of this work. EB, DN, BR, TDS and CR do not have any conflict of interest. DPA’s department has received grant/s from Amgen, Chiesi-Taylor, Lilly, Janssen, Novartis and UCB Biopharma. His research group has received consultancy fees from Astra Zeneca and UCB Biopharma. Amgen, Astellas, Janssen, Synapse Management Partners and UCB Biopharma have funded or supported training programmes organised by DPA’s department.
Figures


References
-
- van Dyck CH, Swanson CJ, Aisen P et al. Lecanemab in early Alzheimer's disease. N Engl J Med 2023; 388: 9–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

