Recent advances in catalytic asymmetric synthesis
- PMID: 38783896
- PMCID: PMC11112575
- DOI: 10.3389/fchem.2024.1398397
Recent advances in catalytic asymmetric synthesis
Abstract
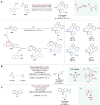
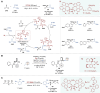
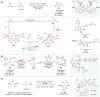
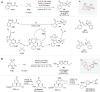
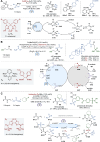
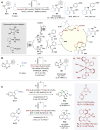
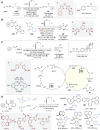
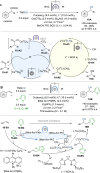
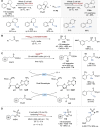
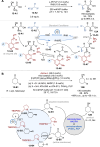
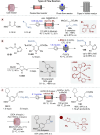
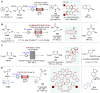
Asymmetric catalysis stands at the forefront of modern chemistry, serving as a cornerstone for the efficient creation of enantiopure chiral molecules characterized by their high selectivity. In this review, we delve into the realm of asymmetric catalytic reactions, which spans various methodologies, each contributing to the broader landscape of the enantioselective synthesis of chiral molecules. Transition metals play a central role as catalysts for a wide range of transformations with chiral ligands such as phosphines, N-heterocyclic carbenes (NHCs), etc., facilitating the formation of chiral C-C and C-X bonds, enabling precise control over stereochemistry. Enantioselective photocatalytic reactions leverage the power of light as a driving force for the synthesis of chiral molecules. Asymmetric electrocatalysis has emerged as a sustainable approach, being both atom-efficient and environmentally friendly, while offering a versatile toolkit for enantioselective reductions and oxidations. Biocatalysis relies on nature's most efficient catalysts, i.e., enzymes, to provide exquisite selectivity, as well as a high tolerance for diverse functional groups under mild conditions. Thus, enzymatic optical resolution, kinetic resolution and dynamic kinetic resolution have revolutionized the production of enantiopure compounds. Enantioselective organocatalysis uses metal-free organocatalysts, consisting of modular chiral phosphorus, sulfur and nitrogen components, facilitating remarkably efficient and diverse enantioselective transformations. Additionally, unlocking traditionally unreactive C-H bonds through selective functionalization has expanded the arsenal of catalytic asymmetric synthesis, enabling the efficient and atom-economical construction of enantiopure chiral molecules. Incorporating flow chemistry into asymmetric catalysis has been transformative, as continuous flow systems provide precise control over reaction conditions, enhancing the efficiency and facilitating optimization. Researchers are increasingly adopting hybrid approaches that combine multiple strategies synergistically to tackle complex synthetic challenges. This convergence holds great promise, propelling the field of asymmetric catalysis forward and facilitating the efficient construction of complex molecules in enantiopure form. As these methodologies evolve and complement one another, they push the boundaries of what can be accomplished in catalytic asymmetric synthesis, leading to the discovery of novel, highly selective transformations which may lead to groundbreaking applications across various industries.
Keywords: C-H activation; asymmetric catalytic synthesis; asymmetric electrocatalysis; asymmetric organocatalysis; asymmetric photocatalysis; biocatalysis; flow chemistry.
Copyright © 2024 Garg, Rendina, Bendale, Akiyama and Ojima.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures



Similar articles
-
Construction of Axially Chiral Compounds via Asymmetric Organocatalysis.Acc Chem Res. 2018 Feb 20;51(2):534-547. doi: 10.1021/acs.accounts.7b00602. Epub 2018 Feb 8. Acc Chem Res. 2018. PMID: 29419282
-
Recent Advances in Asymmetric Organometallic Electrochemical Synthesis (AOES).Acc Chem Res. 2025 Feb 4;58(3):399-414. doi: 10.1021/acs.accounts.4c00656. Epub 2025 Jan 19. Acc Chem Res. 2025. PMID: 39829007
-
Enantioselective "organocatalysis in disguise" by the ligand sphere of chiral metal-templated complexes.Chem Soc Rev. 2021 Sep 7;50(17):9715-9740. doi: 10.1039/d0cs00806k. Epub 2021 Jul 14. Chem Soc Rev. 2021. PMID: 34259242
-
Bifunctional N-Heterocyclic Carbenes Derived from l-Pyroglutamic Acid and Their Applications in Enantioselective Organocatalysis.Acc Chem Res. 2020 Mar 17;53(3):690-702. doi: 10.1021/acs.accounts.9b00635. Epub 2020 Mar 6. Acc Chem Res. 2020. PMID: 32142245 Review.
-
Reticulating Crystalline Porous Materials for Asymmetric Heterogeneous Catalysis.Adv Mater. 2024 Dec 30:e2415574. doi: 10.1002/adma.202415574. Online ahead of print. Adv Mater. 2024. PMID: 39740186 Review.
Cited by
-
Determination of the pKa Value of a Brønsted Acid by 19F NMR Spectroscopy.Magn Reson Chem. 2025 Jan;63(1):17-23. doi: 10.1002/mrc.5485. Epub 2024 Oct 16. Magn Reson Chem. 2025. PMID: 39415461 Free PMC article.
-
Proline-Functionalized Magnetic Nanoparticles as Highly Performing Asymmetric Catalysts.Macromol Rapid Commun. 2024 Dec;45(24):e2400615. doi: 10.1002/marc.202400615. Epub 2024 Sep 11. Macromol Rapid Commun. 2024. PMID: 39259266 Free PMC article.
-
Identification of senescence rejuvenation mechanism of Magnolia officinalis extract including honokiol as a core ingredient.Aging (Albany NY). 2025 Feb 21;17(2):497-523. doi: 10.18632/aging.206207. Epub 2025 Feb 21. Aging (Albany NY). 2025. PMID: 39992207 Free PMC article.
-
Chiral Sodium Glycerophosphate Catalyst for Enantioselective Michael Reactions of Chalcones.Molecules. 2024 Oct 8;29(19):4763. doi: 10.3390/molecules29194763. Molecules. 2024. PMID: 39407691 Free PMC article.
References
-
- Akiyama T., Ojima I. (2022) Catalytic asymmetric synthesis. 4th Edition. USA: John Wiley and Sons.
-
- Albini A., Fagnoni M. (2004). Green chemistry and photochemistry were born at the same time. Green Chem. 6 (1), 1–6. 10.1039/b309592d - DOI
-
- Alfano A. I., Sorato A., Ciogli A., Lange H., Brindisi M. (2023). Enantioselective catalytic Strecker reaction on cyclic (Z)-aldimines in flow: reaction optimization and sustainability aspects. J. Flow Chem. 14, 197–210. 10.1007/s41981-023-00279-9 - DOI
-
- Arnaboldi S., Magni M., Mussini P. R. (2018). Enantioselective selectors for chiral electrochemistry and electroanalysis: stereogenic elements and enantioselection performance. Curr. Opin. Electrochem. 8, 60–72. 10.1016/j.coelec.2018.01.002 - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

