Aβ Oligomer Dissociation Is Catalyzed by Fibril Surfaces
- PMID: 38785363
- PMCID: PMC11157482
- DOI: 10.1021/acschemneuro.4c00127
Aβ Oligomer Dissociation Is Catalyzed by Fibril Surfaces
Abstract
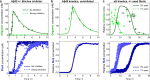
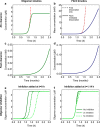
Oligomeric assemblies consisting of only a few protein subunits are key species in the cytotoxicity of neurodegenerative disorders, such as Alzheimer's and Parkinson's diseases. Their lifetime in solution and abundance, governed by the balance of their sources and sinks, are thus important determinants of disease. While significant advances have been made in elucidating the processes that govern oligomer production, the mechanisms behind their dissociation are still poorly understood. Here, we use chemical kinetic modeling to determine the fate of oligomers formed in vitro and discuss the implications for their abundance in vivo. We discover that oligomeric species formed predominantly on fibril surfaces, a broad class which includes the bulk of oligomers formed by the key Alzheimer's disease-associated Aβ peptides, also dissociate overwhelmingly on fibril surfaces, not in solution as had previously been assumed. We monitor this "secondary nucleation in reverse" by measuring the dissociation of Aβ42 oligomers in the presence and absence of fibrils via two distinct experimental methods. Our findings imply that drugs that bind fibril surfaces to inhibit oligomer formation may also inhibit their dissociation, with important implications for rational design of therapeutic strategies for Alzheimer's and other amyloid diseases.
Keywords: Alzheimer’s; dissociation; fibrils; inhibitor; kinetics; oligomer; therapeutic.
Conflict of interest statement
The authors declare the following competing financial interest(s): AJD, GM, SL and TPJK have all previously worked for a company developing therapeutics for neurodegenerative disorders.
Figures


Similar articles
-
Understanding amyloid fibril nucleation and aβ oligomer/drug interactions from computer simulations.Acc Chem Res. 2014 Feb 18;47(2):603-11. doi: 10.1021/ar4002075. Epub 2013 Dec 24. Acc Chem Res. 2014. PMID: 24368046 Review.
-
Mechanism of Secondary Nucleation at the Single Fibril Level from Direct Observations of Aβ42 Aggregation.J Am Chem Soc. 2021 Oct 13;143(40):16621-16629. doi: 10.1021/jacs.1c07228. Epub 2021 Sep 28. J Am Chem Soc. 2021. PMID: 34582216
-
Off-pathway oligomers of α-synuclein and Aβ inhibit secondary nucleation of α-synuclein amyloid fibrils.J Mol Biol. 2025 May 15;437(10):169048. doi: 10.1016/j.jmb.2025.169048. Epub 2025 Feb 25. J Mol Biol. 2025. PMID: 40015369
-
Proliferation of amyloid-β42 aggregates occurs through a secondary nucleation mechanism.Proc Natl Acad Sci U S A. 2013 Jun 11;110(24):9758-63. doi: 10.1073/pnas.1218402110. Epub 2013 May 23. Proc Natl Acad Sci U S A. 2013. PMID: 23703910 Free PMC article.
-
Elucidating the Structures of Amyloid Oligomers with Macrocyclic β-Hairpin Peptides: Insights into Alzheimer's Disease and Other Amyloid Diseases.Acc Chem Res. 2018 Mar 20;51(3):706-718. doi: 10.1021/acs.accounts.7b00554. Epub 2018 Mar 6. Acc Chem Res. 2018. PMID: 29508987 Free PMC article. Review.
Cited by
-
On the reversibility of amyloid fibril formation.Biophys Rev (Melville). 2025 Jan 29;6(1):011303. doi: 10.1063/5.0236947. eCollection 2025 Mar. Biophys Rev (Melville). 2025. PMID: 39973975 Free PMC article. Review.
-
Ultrasensitive Assays Detect Different Conformations of Plasma β Amyloids.ACS Omega. 2025 Feb 11;10(7):7256-7263. doi: 10.1021/acsomega.4c10879. eCollection 2025 Feb 25. ACS Omega. 2025. PMID: 40028141 Free PMC article.
-
Detecting and Tracking β-Amyloid Oligomeric Forms and Dynamics In Vitro by a High-Sensitivity Fluorescent-Based Assay.ACS Chem Neurosci. 2024 Dec 18;15(24):4383-4389. doi: 10.1021/acschemneuro.4c00312. Epub 2024 Nov 29. ACS Chem Neurosci. 2024. PMID: 39611283 Free PMC article.
-
Accelerated Alzheimer's Aβ-42 secondary nucleation chronologically visualized on fibril surfaces.Sci Adv. 2024 Oct 25;10(43):eadp5059. doi: 10.1126/sciadv.adp5059. Epub 2024 Oct 25. Sci Adv. 2024. PMID: 39454002 Free PMC article.
-
An approach to predict and inhibit Amyloid Beta dimerization pattern in Alzheimer's disease.Toxicol Rep. 2024 Dec 28;14:101879. doi: 10.1016/j.toxrep.2024.101879. eCollection 2025 Jun. Toxicol Rep. 2024. PMID: 39867516 Free PMC article.
References
-
- Mcnaught A. D.; Wilkinson A.. IUPAC. Compendium of Chemical Terminology, (The “Gold Book”), 2nd ed.; Blackwell Scientific Publications, 1997.
-
- Guerrero-Muñoz M. J.; Castillo-Carranza D. L.; Krishnamurthy S.; Paulucci-Holthauzen A. A.; Sengupta U.; Lasagna-Reeves C. A.; Ahmad Y.; Jackson G. R.; Kayed R. Amyloid-β oligomers as a template for secondary amyloidosis in Alzheimer’s disease. Neurobiol. Dis. 2014, 71, 14–23. 10.1016/j.nbd.2014.08.008. - DOI - PubMed
-
- Conway K. A.; Lee S. J.; Rochet J. C.; Ding T. T.; Williamson R. E.; Lansbury P. T. Acceleration of oligomerization, not fibrillization, is a shared property of both α-synuclein mutations linked to early-onset Parkinson’s disease: implications for pathogenesis and therapy. Proc. Natl. Acad. Sci. U.S.A. 2000, 97, 571–576. 10.1073/pnas.97.2.571. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

