The Role of TLR-2 in Lethal COVID-19 Disease Involving Medullary and Resident Lung Megakaryocyte Up-Regulation in the Microthrombosis Mechanism
- PMID: 38786077
- PMCID: PMC11120208
- DOI: 10.3390/cells13100854
The Role of TLR-2 in Lethal COVID-19 Disease Involving Medullary and Resident Lung Megakaryocyte Up-Regulation in the Microthrombosis Mechanism
Abstract
Patients with COVID-19 have coagulation and platelet disorders, with platelet alterations and thrombocytopenia representing negative prognostic parameters associated with severe forms of the disease and increased lethality.
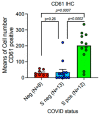
Methods: The aim of this study was to study the expression of platelet glycoprotein IIIa (CD61), playing a critical role in platelet aggregation, together with TRL-2 as a marker of innate immune activation.
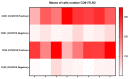
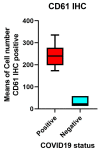
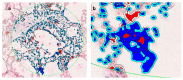
Results: A total of 25 patients were investigated, with the majority (24/25, 96%) having co-morbidities and dying from a fatal form of SARS-CoV-2(+) infection (COVID-19+), with 13 men and 12 females ranging in age from 45 to 80 years. When compared to a control group of SARS-CoV-2 (-) negative lungs (COVID-19-), TLR-2 expression was up-regulated in a subset of patients with deadly COVID-19 fatal lung illness. The proportion of Spike-1 (+) patients found by PCR and ISH correlates to the proportion of Spike-S1-positive cases as detected by digital pathology examination. Furthermore, CD61 expression was considerably higher in the lungs of deceased patients. In conclusion, we demonstrate that innate immune prolonged hyperactivation is related to platelet/megakaryocyte over-expression in the lung.
Conclusions: Microthrombosis in deadly COVID-19+ lung disease is associated with an increase in the number of CD61+ platelets and megakaryocytes in the pulmonary interstitium, as well as their functional activation; this phenomenon is associated with increased expression of innate immunity TLR2+ cells, which binds the SARS-CoV-2 E protein, and significantly with the persistence of the Spike-S1 viral sequence.
Keywords: ARDS; CD61; SARS-CoV-2; TRL-2; TRLs; lethal COVID-19; lung disease; megakaryocytes; micro-thrombosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- D’Atri L.P., Schattner M. Platelet toll-like receptors in thromboinflammation. Front. Biosci. 2017;22:1867–1883. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

