Inhibition of Candida albicans Biofilm Formation and Attenuation of Its Virulence by Liriope muscari
- PMID: 38786162
- PMCID: PMC11117302
- DOI: 10.3390/antibiotics13050434
Inhibition of Candida albicans Biofilm Formation and Attenuation of Its Virulence by Liriope muscari
Abstract
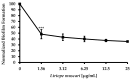
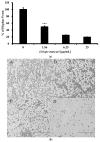
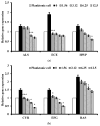
(1) Background: Although Candida albicans accounts for the majority of fungal infections, therapeutic options are limited and require alternative antifungal agents with new targets; (2) Methods: A biofilm formation assay with RPMI1640 medium was performed with Liriope muscari extract. A combination antifungal assay, dimorphic transition assay, and adhesion assay were performed under the biofilm formation condition to determine the anti-biofilm formation effect. qRT-PCR analysis was accomplished to confirm changes in gene expression; (3) Results: L. muscari extract significantly reduces biofilm formation by 51.65% at 1.56 μg/mL use and therefore increases susceptibility to miconazole. L. muscari extract also inhibited the dimorphic transition of Candida; nearly 50% of the transition was inhibited when 1.56 μg/mL of the extract was treated. The extract of L. muscari inhibited the expression of genes related to hyphal development and extracellular matrix of 34.4% and 36.0%, respectively, as well as genes within the Ras1-cAMP-PKA, Cph2-Tec1, and MAP kinase signaling pathways of 25.58%, 7.1% and 15.8%, respectively, at 1.56 μg/mL of L. muscari extract treatment; (4) Conclusions: L. muscari extract significantly reduced Candida biofilm formation, which lead to induced antifungal susceptibility to miconazole. It suggests that L. muscari extract is a promising anti-biofilm candidate of Candida albicans since the biofilm formation of Candida albicans is an excellent target for candidiasis regulation.
Keywords: Candida albicans; Liriope muscari; adherence assay; antifungal agent; biofilm formation; dimorphic transition.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




References
Grants and funding
LinkOut - more resources
Full Text Sources

