Neutrophils disrupt B-1a cell homeostasis by targeting Siglec-G to exacerbate sepsis
- PMID: 38789529
- PMCID: PMC11214631
- DOI: 10.1038/s41423-024-01165-7
Neutrophils disrupt B-1a cell homeostasis by targeting Siglec-G to exacerbate sepsis
Abstract
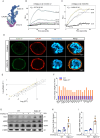
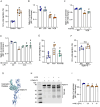
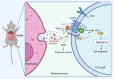
B-1a cells, an innate-like cell population, are crucial for pathogen defense and the regulation of inflammation through their release of natural IgM and IL-10. In sepsis, B-1a cell numbers are decreased in the peritoneal cavity as they robustly migrate to the spleen. Within the spleen, migrating B-1a cells differentiate into plasma cells, leading to alterations in their original phenotype and functionality. We discovered a key player, sialic acid-binding immunoglobulin-like lectin-G (Siglec-G), which is expressed predominantly on B-1a cells and negatively regulates B-1a cell migration to maintain homeostasis. Siglec-G interacts with CXCR4/CXCL12 to modulate B-1a cell migration. Neutrophils aid B-1a cell migration via neutrophil elastase (NE)-mediated Siglec-G cleavage. Human studies revealed increased NE expression in septic patients. We identified an NE cleavage sequence in silico, leading to the discovery of a decoy peptide that protects Siglec-G, preserves peritoneal B-1a cells, reduces inflammation, and enhances sepsis survival. The role of Siglec-G in inhibiting B-1a cell migration to maintain their inherent phenotype and function is compromised by NE in sepsis, offering valuable insights into B-1a cell homeostasis. Employing a small decoy peptide to prevent NE-mediated Siglec-G cleavage has emerged as a promising strategy to sustain peritoneal B-1a cell homeostasis, alleviate inflammation, and ultimately improve outcomes in sepsis patients.
Keywords: B-1a cells; CXCL12; CXCR4; Neutrophil elastase; Neutrophils; Sepsis; Siglec-G.
© 2024. The Author(s), under exclusive licence to CSI and USTC.
Conflict of interest statement
The authors declare no competing interests.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

