Drug Integrating Amphiphilic Nano-Assemblies: 2. Spatiotemporal Distribution within Inflammation Sites
- PMID: 38794314
- PMCID: PMC11124943
- DOI: 10.3390/pharmaceutics16050652
Drug Integrating Amphiphilic Nano-Assemblies: 2. Spatiotemporal Distribution within Inflammation Sites
Abstract
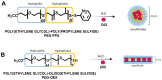
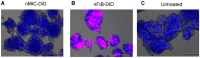
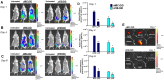
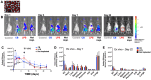
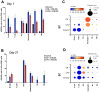
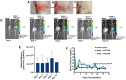
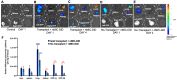
The need for chronic systemic immunosuppression, which is associated with unavoidable side-effects, greatly limits the applicability of allogeneic cell transplantation for regenerative medicine applications including pancreatic islet cell transplantation to restore insulin production in type 1 diabetes (T1D). Cell transplantation in confined sites enables the localized delivery of anti-inflammatory and immunomodulatory drugs to prevent graft loss by innate and adaptive immunity, providing an opportunity to achieve local effects while minimizing unwanted systemic side effects. Nanoparticles can provide the means to achieve the needed localized and sustained drug delivery either by graft targeting or co-implantation. Here, we evaluated the potential of our versatile platform of drug-integrating amphiphilic nanomaterial assemblies (DIANAs) for targeted drug delivery to an inflamed site model relevant for islet transplantation. We tested either passive targeting of intravenous administered spherical nanomicelles (nMIC; 20-25 nm diameter) or co-implantation of elongated nanofibrils (nFIB; 5 nm diameter and >1 μm length). To assess the ability of nMIC and nFIB to target an inflamed graft site, we used a lipophilic fluorescent cargo (DiD and DiR) and evaluated the in vivo biodistribution and cellular uptake in the graft site and other organs, including draining and non-draining lymph nodes, after systemic administration (nMIC) and/or graft co-transplantation (nFIB) in mice. Localized inflammation was generated either by using an LPS injection or by using biomaterial-coated islet-like bead implantation in the subcutaneous site. A cell transplant inflammation model was used as well to test nMIC- and nFIB-targeted biodistribution. We found that nMIC can reach the inflamed site after systemic administration, while nFIB remains localized for several days after co-implantation. We confirmed that DIANAs are taken up by different immune cell populations responsible for graft inflammation. Therefore, DIANA is a useful approach for targeted and/or localized delivery of immunomodulatory drugs to decrease innate and adaptive immune responses that cause graft loss after transplantation of therapeutic cells.
Keywords: block-copolymers; cell transplantation; drug delivery; local immunomodulation; nanoparticles; self-assembling.
Conflict of interest statement
The authors declare the following competing financial interest(s): D.V. is inventor on patents and patent applications related to these nanomaterials. All other authors declare no competing financial interests. A.A.T. is an inventor of intellectual property used in the study for the conformal coating encapsulation, licensed to Sernova, and may gain royalties from future commercialization of the technology.
Figures


Similar articles
-
Drug-Integrating Amphiphilic Nanomaterial Assemblies: 1. Spatiotemporal control of cyclosporine delivery and activity using nanomicelles and nanofibrils.J Control Release. 2021 Jan 10;329:955-970. doi: 10.1016/j.jconrel.2020.10.026. Epub 2020 Oct 18. J Control Release. 2021. PMID: 33086102 Free PMC article.
-
Designing biomaterials for the modulation of allogeneic and autoimmune responses to cellular implants in Type 1 Diabetes.Acta Biomater. 2021 Oct 1;133:87-101. doi: 10.1016/j.actbio.2021.05.039. Epub 2021 Jun 5. Acta Biomater. 2021. PMID: 34102338 Free PMC article. Review.
-
Immunosuppressive PLGA TGF-β1 Microparticles Induce Polyclonal and Antigen-Specific Regulatory T Cells for Local Immunomodulation of Allogeneic Islet Transplants.Front Immunol. 2021 May 27;12:653088. doi: 10.3389/fimmu.2021.653088. eCollection 2021. Front Immunol. 2021. PMID: 34122410 Free PMC article.
-
Evaluation of biomaterial scaffold delivery of IL-33 as a localized immunomodulatory agent to support cell transplantation in adipose tissue.J Immunol Regen Med. 2018 Mar;1:1-12. doi: 10.1016/j.regen.2018.01.003. Epub 2018 Feb 22. J Immunol Regen Med. 2018. PMID: 29869643 Free PMC article.
-
Local Immunomodulatory Strategies to Prevent Allo-Rejection in Transplantation of Insulin-Producing Cells.Adv Sci (Weinh). 2021 Sep;8(17):e2003708. doi: 10.1002/advs.202003708. Epub 2021 Jul 14. Adv Sci (Weinh). 2021. PMID: 34258870 Free PMC article. Review.
Cited by
-
Challenges in the diagnosis of primary squamous cell carcinoma of the prostate: a case report and literature review.Front Surg. 2025 Jul 17;12:1532669. doi: 10.3389/fsurg.2025.1532669. eCollection 2025. Front Surg. 2025. PMID: 40746833 Free PMC article.
-
From Edmonton to Lantidra and beyond: immunoengineering islet transplantation to cure type 1 diabetes.Front Transplant. 2025 Mar 20;4:1514956. doi: 10.3389/frtra.2025.1514956. eCollection 2025. Front Transplant. 2025. PMID: 40182604 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

