Targeted next-generation sequencing to diagnose drug-resistant tuberculosis: a systematic review and meta-analysis
- PMID: 38795712
- PMCID: PMC11881551
- DOI: 10.1016/S1473-3099(24)00263-9
Targeted next-generation sequencing to diagnose drug-resistant tuberculosis: a systematic review and meta-analysis
Abstract
Background: Targeted next-generation sequencing (NGS) can rapidly and simultaneously detect mutations associated with resistance to tuberculosis drugs across multiple gene targets. The use of targeted NGS to diagnose drug-resistant tuberculosis, as described in publicly available data, has not been comprehensively reviewed. We aimed to identify targeted NGS assays that diagnose drug-resistant tuberculosis, determine how widely this technology has been used, and assess the diagnostic accuracy of these assays.
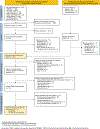
Methods: In this systematic review and meta-analysis, we searched MEDLINE, Embase, Cochrane Library, Web of Science Core Collection, Global Index Medicus, Google Scholar, ClinicalTrials.gov, and the WHO International Clinical Trials Registry Platform for published and unpublished reports on targeted NGS for drug-resistant tuberculosis from Jan 1, 2005, to Oct 14, 2022, with updates to our search in Embase and Google Scholar until Feb 13, 2024. Studies eligible for the systematic review described targeted NGS approaches to predict drug resistance in Mycobacterium tuberculosis infections using primary samples, reference strain collections, or cultured isolates from individuals with presumed or confirmed tuberculosis. Our search had no limitations on study type or language, although only reports in English, German, and French were screened for eligibility. For the meta-analysis, we included test accuracy studies that used any reference standard, and we assessed risk of bias using the Quality Assessment of Diagnostic Accuracy Studies-2 tool. The primary outcomes for the meta-analysis were sensitivity and specificity of targeted NGS to diagnose drug-resistant tuberculosis compared to phenotypic and genotypic drug susceptibility testing. We used a Bayesian bivariate model to generate summary receiver operating characteristic plots and diagnostic accuracy measures, overall and stratified by drug and sample type. This study is registered with PROSPERO, CRD42022368707.
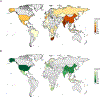
Findings: We identified and screened 2920 reports, of which 124 were eligible for our systematic review, including 37 review articles and 87 reports of studies collecting samples for targeted NGS. Sequencing was mainly done in the USA (14 [16%] of 87), western Europe (ten [11%]), India (ten [11%]), and China (nine [10%]). We included 24 test accuracy studies in the meta-analysis, in which 23 different tuberculosis drugs or drug groups were assessed, covering first-line drugs, injectable drugs, and fluoroquinolones and predominantly comparing targeted NGS with phenotypic drug susceptibility testing. The combined sensitivity of targeted NGS across all drugs was 94·1% (95% credible interval [CrI] 90·9-96·3) and specificity was 98·1% (97·0-98·9). Sensitivity for individual drugs ranged from 76·5% (52·5-92·3) for capreomycin to 99·1% (98·3-99·7) for rifampicin; specificity ranged from 93·1% (88·0-96·3) for ethambutol to 99·4% (98·3-99·8) for amikacin. Diagnostic accuracy was similar for primary clinical samples and culture isolates overall and for rifampicin, isoniazid, ethambutol, streptomycin, and fluoroquinolones, and similar after excluding studies at high risk of bias (overall sensitivity 95·2% [95% CrI 91·7-97·1] and specificity 98·6% [97·4-99·3]).
Interpretation: Targeted NGS is highly sensitive and specific for detecting drug resistance across panels of tuberculosis drugs and can be performed directly on clinical samples. There is a paucity of data on performance for some currently recommended drugs. The barriers preventing the use of targeted NGS to diagnose drug-resistant tuberculosis in high-burden countries need to be addressed.
Funding: National Institutes of Allergy and Infectious Diseases and Swiss National Science Foundation.
Copyright © 2024 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests We declare no competing interests.
Figures
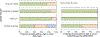

References
-
- World Health Organization. Technical manual for drug susceptibility testing of medicines used in the treatment of tuberculosis. Geneva: World Health Organization, 2018.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

