Discriminating orientation information with phase consistency in alpha and low-gamma frequency bands: an EEG study
- PMID: 38796618
- PMCID: PMC11127946
- DOI: 10.1038/s41598-024-62934-y
Discriminating orientation information with phase consistency in alpha and low-gamma frequency bands: an EEG study
Abstract
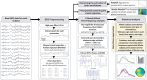
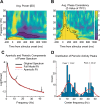
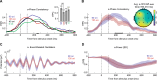
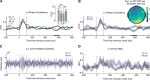
Recent studies suggest that noninvasive imaging methods (EEG, MEG) in the human brain scalp can decode the content of visual features information (orientation, color, motion, etc.) in Visual-Working Memory (VWM). Previous work demonstrated that with the sustained low-frequency Event-Related Potential (ERP under 6 Hz) of scalp EEG distributions, it is possible to accurately decode the content of orientation information in VWM during the delay interval. In addition, previous studies showed that the raw data captured by a combination of the occi-parietal electrodes could be used to decode the orientation. However, it is unclear whether the orientation information is available in other frequency bands (higher than 6 Hz) or whether this information is feasible with fewer electrodes. Furthermore, the exploration of orientation information in the phase values of the signal has not been well-addressed. In this study, we propose that orientation information is also accessible through the phase consistency of the occipital region in the alpha band frequency. Our results reveal a significant difference between orientations within 200 ms after stimulus offset in early visual sensory processing, with no apparent effect in power and Event-Related Oscillation (ERO) during this period. Additionally, in later periods (420-500 ms after stimulus offset), a noticeable difference is observed in the phase consistency of low gamma-band activity in the occipital area. Importantly, our findings suggest that phase consistency between trials of the orientation feature in the occipital alpha and low gamma-band can serve as a measure to obtain orientation information in VWM. Furthermore, the study demonstrates that phase consistency in the alpha and low gamma band can reflect the distribution of orientation-selective neuron numbers in the four main orientations in the occipital area.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
MeSH terms
LinkOut - more resources
Full Text Sources

