Oxygen self-sufficient nanodroplet composed of fluorinated polymer for high-efficiently PDT eradicating oral biofilm
- PMID: 38800565
- PMCID: PMC11126933
- DOI: 10.1016/j.mtbio.2024.101091
Oxygen self-sufficient nanodroplet composed of fluorinated polymer for high-efficiently PDT eradicating oral biofilm
Abstract
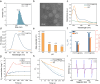
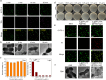
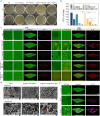
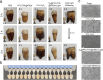
Oral biofilm is the leading cause of dental caries, which is difficult to completely eradicate because of the complicated biofilm structure. What's more, the hypoxia environment of biofilm and low water-solubility of conventional photosensitizers severely restrict the therapeutic effect of photodynamic therapy (PDT) for biofilm. Although conventional photosensitizers could be loaded in nanocarriers, it has reduced PDT effect because of aggregation-caused quenching (ACQ) phenomenon. In this study, we fabricated an oxygen self-sufficient nanodroplet (PFC/TPA@FNDs), which was composed of fluorinated-polymer (FP), perfluorocarbons (PFC) and an aggregation-induced emission (AIE) photosensitizer (Triphenylamine, TPA), to eradicate oral bacterial biofilm and whiten tooth. Fluorinated-polymer was synthesized by polymerizing (Dimethylamino)ethyl methacrylate, fluorinated monomer and 1-nonanol monomer. The nanodroplets could be protonated and behave strong positive charge under bacterial biofilm acid environment promoting nanodroplets deeply penetrating biofilm. More importantly, the nanodroplets had extremely high PFC and oxygen loading efficacy because of the hydrophobic affinity between fluorinated-polymer and PFC to relieve the hypoxia environment and enhance PDT effect. Additionally, compared with conventional ACQ photosensitizers loaded system, PFC/TPA@FNDs could behave superior PDT effect to ablate oral bacterial biofilm under light irradiation due to the unique AIE effect. In vivo caries animal model proved the nanodroplets could reduce dental caries area without damaging tooth structure. Ex vivo tooth whitening assay also confirmed the nanodroplets had similar tooth whitening ability compared with commercial tooth whitener H2O2, while did not disrupt the surface microstructure of tooth. This oxygen self-sufficient nanodroplet provides an alternative visual angle for oral biofilm eradication in biomedicine.
Keywords: AIE photosensitizer; Dental caries; Fluorinated-polymer; Nanodroplets; Oral-biofilm.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures




Similar articles
-
Photoacoustic nanodroplets for oxygen enhanced photodynamic therapy of cancer.Photoacoustics. 2021 Sep 27;25:100306. doi: 10.1016/j.pacs.2021.100306. eCollection 2022 Mar. Photoacoustics. 2021. PMID: 34917471 Free PMC article.
-
Rational collaborative ablation of bacterial biofilms ignited by physical cavitation and concurrent deep antibiotic release.Biomaterials. 2020 Dec;262:120341. doi: 10.1016/j.biomaterials.2020.120341. Epub 2020 Aug 28. Biomaterials. 2020. PMID: 32911255
-
Oxygen self-sufficient fluorinated polypeptide nanoparticles for NIR imaging-guided enhanced photodynamic therapy.J Mater Chem B. 2018 Apr 21;6(15):2323-2331. doi: 10.1039/c8tb00493e. Epub 2018 Apr 3. J Mater Chem B. 2018. PMID: 32254571
-
AIE-Active Photosensitizers: Manipulation of Reactive Oxygen Species Generation and Applications in Photodynamic Therapy.Biosensors (Basel). 2022 May 18;12(5):348. doi: 10.3390/bios12050348. Biosensors (Basel). 2022. PMID: 35624649 Free PMC article. Review.
-
Reviewing the evolutive ACQ-to-AIE transformation of photosensitizers for phototheranostics.Luminescence. 2023 Dec 26. doi: 10.1002/bio.4655. Online ahead of print. Luminescence. 2023. PMID: 38148620 Review.
Cited by
-
Animal models of oral infectious diseases.Front Oral Health. 2025 Apr 17;6:1571492. doi: 10.3389/froh.2025.1571492. eCollection 2025. Front Oral Health. 2025. PMID: 40351786 Free PMC article. Review.
-
Advanced biomaterials for targeting mature biofilms in periodontitis therapy.Bioact Mater. 2025 Feb 27;48:474-492. doi: 10.1016/j.bioactmat.2025.02.026. eCollection 2025 Jun. Bioact Mater. 2025. PMID: 40093304 Free PMC article. Review.
References
-
- Peres M.A., Macpherson L.M.D., Weyant R.J., Daly B., Venturelli R., Mathur M.R., Listl S., Celeste R.K., Guarnizo-Herreno C.C., Kearns C., Benzian H., Allison P., Watt R.G. Oral diseases: a global public health challenge. Lancet. 2019;394(10194):249–260. doi: 10.1016/S0140-6736(19)31146-8. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

