Regioselective Palladium-Catalyzed Chain-Growth Allylic Amination Polymerization of Vinyl Aziridines
- PMID: 38801413
- PMCID: PMC11977030
- DOI: 10.1021/jacs.4c02599
Regioselective Palladium-Catalyzed Chain-Growth Allylic Amination Polymerization of Vinyl Aziridines
Abstract
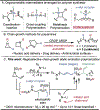
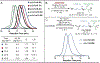
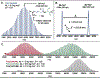
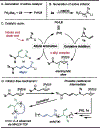
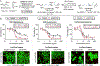
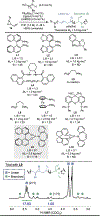
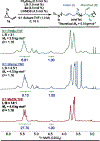
Organometallic-mediated chain growth polymerization of readily accessible chemical building blocks is responsible for important commercial and technological advances in polymer science, but the incorporation of heteroatoms into the polymer backbone through these mechanisms remains a challenge. Transition metal π-allyl complexes are well-developed organometallic intermediates for carbon-heteroatom bond formation in small-molecule catalysis yet remain underexplored in polymer science. Here, we developed a regioselective palladium-phosphoramidite-catalyzed chain-growth allylic amination polymerization of vinyl aziridines for the synthesis of novel nitrogen-rich polymers via ambiphilic π-allyl complexes. The polymerization accessed a linear microstructure with four carbons between each nitrogen, which is challenging to achieve through other chain-growth polymerization approaches. The highly regioselective allylic amination polymerization demonstrated the characteristics of a controlled polymerization and was able to achieve molar masses exceeding 20 kg mol-1 with low dispersities (D̵ < 1.3). The identification of the polymer structure and well-defined chain ends were supported by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry, and chain extension experiments demonstrate opportunities for building more complex materials from this method. A Hammett study was performed to understand the role of the catalyst and monomer structure on regioselectivity, and the data supported a mechanism wherein regioselectivity was primarily controlled by the ligand-metal complex. Postpolymerization desulfonylation provided access to a novel polyamine that demonstrated broad anticancer activity in vitro, which highlights the benefits of unlocking novel polyamine microstructures through regioselective chain-growth allylic amination polymerization.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
New insights into the mechanism of palladium-catalyzed allylic amination.J Am Chem Soc. 2005 Dec 14;127(49):17516-29. doi: 10.1021/ja055288c. J Am Chem Soc. 2005. PMID: 16332104
-
Palladium-Catalyzed Regioselective and Stereospecific Ring-Opening Cross-Coupling of Aziridines: Experimental and Computational Studies.Acc Chem Res. 2020 Aug 18;53(8):1686-1702. doi: 10.1021/acs.accounts.0c00395. Epub 2020 Aug 6. Acc Chem Res. 2020. PMID: 32786337
-
Mechanistically driven development of iridium catalysts for asymmetric allylic substitution.Acc Chem Res. 2010 Dec 21;43(12):1461-75. doi: 10.1021/ar100047x. Epub 2010 Sep 28. Acc Chem Res. 2010. PMID: 20873839 Free PMC article.
-
Palladium-catalyzed allylic C-H bond functionalization of olefins.Top Curr Chem. 2010;292:195-209. doi: 10.1007/128_2009_16. Top Curr Chem. 2010. PMID: 21500407 Review.
-
Non-Bonding Interactions Enable the Selective Formation of Branched Products in Palladium-Catalyzed Allylic Substitution Reactions.Chem Asian J. 2018 Sep 4;13(17):2174-2183. doi: 10.1002/asia.201800496. Epub 2018 Jul 4. Chem Asian J. 2018. PMID: 29971957 Review.
Cited by
-
Stereoconvergent Chain-Growth Polymerization.ACS Cent Sci. 2025 May 5;11(5):797-804. doi: 10.1021/acscentsci.5c00239. eCollection 2025 May 28. ACS Cent Sci. 2025. PMID: 40454343 Free PMC article.
-
Controlled chain-growth polymerization via propargyl/allenyl palladium intermediates.Nat Commun. 2025 Mar 13;16(1):2506. doi: 10.1038/s41467-025-57723-8. Nat Commun. 2025. PMID: 40082432 Free PMC article.
References
-
- Grubbs RB; Grubbs RH 50th Anniversary Perspective: Living Polymerization—Emphasizing the Molecule in Macromolecules. Macromolecules 2017, 50 (18), 6979–6997.
-
- Demirors M The History of Polyethylene. 100+ Years of Plastics. Leo Baekeland and Beyond; ACS Symposium Series; American Chemical Society, 2011; Vol. 1080; pp 115–145.
-
- Swager TM 50th Anniversary Perspective: Conducting/Semiconducting Conjugated Polymers. A Personal Perspective on the Past and the Future. Macromolecules 2017, 50 (13), 4867–4886.
-
- Grubbs RH; Tumas W Polymer Synthesis and Organotransition Metal Chemistry. Science 1989, 243 (4893), 907–915. - PubMed
-
- Kumar S; Dholakiya BZ; Jangir R Role of Organometallic Complexes in Olefin Polymerization: A Review Report. J. Organomet. Chem. 2021, 953, 122066.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

