Immunomodulatory extracellular matrix hydrogel induces tissue regeneration in a model of partial glossectomy
- PMID: 38803824
- PMCID: PMC11128682
- DOI: 10.1016/j.bioactmat.2024.05.001
Immunomodulatory extracellular matrix hydrogel induces tissue regeneration in a model of partial glossectomy
Abstract
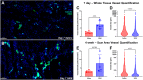
While oropharyngeal cancer treatment regimens, including surgical resection, irradiation, and chemotherapy, are effective at removing tumors, they lead to muscle atrophy, denervation, and fibrosis, contributing to the pathogenesis of oropharyngeal dysphagia - difficulty swallowing. Current standard of care of rehabilitative tongue strengthening and swallowing exercises is ineffective. Here, we evaluate an alternative approach utilizing an acellular and injectable biomaterial to preserve muscle content and reduce fibrosis of the tongue after injury. Skeletal muscle extracellular matrix (SKM) hydrogel is fabricated from decellularized porcine skeletal muscle tissue. A partial glossectomy injury in the rat is used to induce tongue fibrosis, and SKM hydrogels along with saline controls are injected into the site of scarring two weeks after injury. Tissues are harvested at 3 and 7 days post-injection for gene expression and immunohistochemical analyses, and at 4 weeks post-injection to evaluate histomorphological properties. SKM hydrogel reduces scar formation and improves muscle regeneration at the site of injury compared to saline. SKM additionally modulates the immune response towards an anti-inflammatory phenotype. This study demonstrates the immunomodulatory and tissue-regenerative capacity of an acellular and minimally invasive ECM hydrogel in a rodent model of tongue injury.
Keywords: Biomaterial; Dysphagia; Extracellular matrix; Hydrogel; Immunomodulation; Partial glossectomy; Skeletal muscle regeneration.
© 2024 The Authors.
Conflict of interest statement
Dr. Christman is a co-founder, board member, consultant, and holds equity interest in VentrixBio, Inc. and Karios Technologies, Inc.
Figures








References
-
- B. R. Pauloski, n.d., DOI 10.1016/j.pmr.2008.05.010.
-
- García-Peris P., Parón L., Velasco C., de la Cuerda C., Camblor M., Bretón I., Herencia H., Verdaguer J., Navarro C., Clave P. Clin. Nutr. 2007;26:710. - PubMed
-
- Nguyen N.P., Moltz C.C., Frank C., Vos P., Smith H.J., Karlsson U., Dutta S., Midyett F.A., Barloon J., Sallah S. Ann. Oncol. 2004;15:383. - PubMed

