ATP1A3 regulates protein synthesis for mitochondrial stability under heat stress
- PMID: 38804677
- PMCID: PMC11247502
- DOI: 10.1242/dmm.050574
ATP1A3 regulates protein synthesis for mitochondrial stability under heat stress
Abstract
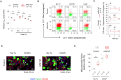
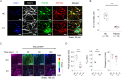
Pathogenic variants in ATP1A3, the gene encoding the α3 subunit of the Na+/K+-ATPase, cause alternating hemiplegia of childhood (AHC) and related disorders. Impairments in Na+/K+-ATPase activity are associated with the clinical phenotype. However, it remains unclear whether additional mechanisms are involved in the exaggerated symptoms under stressed conditions in patients with AHC. We herein report that the intracellular loop (ICL) of ATP1A3 interacted with RNA-binding proteins, such as Eif4g (encoded by Eif4g1), Pabpc1 and Fmrp (encoded by Fmr1), in mouse Neuro2a cells. Both the siRNA-mediated depletion of Atp1a3 and ectopic expression of the p.R756C variant of human ATP1A3-ICL in Neuro2a cells resulted in excessive phosphorylation of ribosomal protein S6 (encoded by Rps6) and increased susceptibility to heat stress. In agreement with these findings, induced pluripotent stem cells (iPSCs) from a patient with the p.R756C variant were more vulnerable to heat stress than control iPSCs. Neurons established from the patient-derived iPSCs showed lower calcium influxes in responses to stimulation with ATP than those in control iPSCs. These data indicate that inefficient protein synthesis contributes to the progressive and deteriorating phenotypes in patients with the p.R756C variant among a variety of ATP1A3-related disorders.
Keywords: Alternating hemiplegia of childhood; Heat stress; Induced pluripotent stem cells (iPSCs); Interaction; Mitochondria; Na+/K+-ATPase α3 subunit (ATP1A3); Protein synthesis.
© 2024. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures






References
-
- Arystarkhova, E., Haq, I. U., Luebbert, T., Mochel, F., Saunders-Pullman, R., Bressman, S. B., Feschenko, P., Salazar, C., Cook, J. F., Demarest, S.et al. (2019). Factors in the disease severity of ATP1A3 mutations: Impairment, misfolding, and allele competition. Neurobiol. Dis. 132, 104577. 10.1016/j.nbd.2019.104577 - DOI - PMC - PubMed
-
- Arystarkhova, E., Toustrup-Jensen, M. S., Holm, R., Ko, J. K., Lee, K. E., Feschenko, P., Ozelius, L. J., Brashear, A., Vilsen, B. and Sweadner, K. J. (2023). Temperature instability of a mutation at a multidomain junction in Na,K-ATPase isoform ATP1A3 (p.Arg756His) produces a fever-induced neurological syndrome. J. Biol. Chem. 299, 102758. 10.1016/j.jbc.2022.102758 - DOI - PMC - PubMed
-
- Barwe, S. P., Anilkumar, G., Moon, S. Y., Zheng, Y., Whitelegge, J. P., Rajasekaran, S. A. and Rajasekaran, A. K. (2005). Novel role for Na,K-ATPase in phosphatidylinositol 3-kinase signaling and suppression of cell motility. Mol. Biol. Cell 16, 1082-1094. 10.1091/mbc.e04-05-0427 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

