Species Difference? Bovine, Trout, and Human Plasma Protein Binding of Per- and Polyfluoroalkyl Substances
- PMID: 38804966
- PMCID: PMC11171458
- DOI: 10.1021/acs.est.3c10824
Species Difference? Bovine, Trout, and Human Plasma Protein Binding of Per- and Polyfluoroalkyl Substances
Abstract
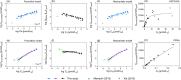
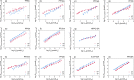
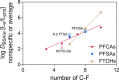
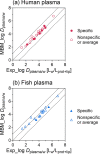
Per- and polyfluoroalkyl substances (PFAS) strongly bind to proteins and lipids in blood, which govern their accumulation and distribution in organisms. Understanding the plasma binding mechanism and species differences will facilitate the quantitative in vitro-to-in vivo extrapolation and improve risk assessment of PFAS. We studied the binding mechanism of 16 PFAS to bovine serum albumin (BSA), trout, and human plasma using solid-phase microextraction. Binding of anionic PFAS to BSA and human plasma was found to be highly concentration-dependent, while trout plasma binding was linear for the majority of the tested PFAS. At a molar ratio of PFAS to protein ν < 0.1 molPFAS/molprotein, the specific protein binding of anionic PFAS dominated their human plasma binding. This would be the scenario for physiological conditions (ν < 0.01), whereas in in vitro assays, PFAS are often dosed in excess (ν > 1) and nonspecific binding becomes dominant. BSA was shown to serve as a good surrogate for human plasma. As trout plasma contains more lipids, the nonspecific binding to lipids affected the affinities of PFAS for trout plasma. Mass balance models that are parameterized with the protein-water and lipid-water partitioning constants (chemical characteristics), as well as the protein and lipid contents of the plasma (species characteristics), were successfully used to predict the binding to human and trout plasma.
Keywords: PFAS; plasma binding mechanism; proteins and lipids; solid-phase microextraction; specific and nonspecific protein binding.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Quantitative cross-species comparison of serum albumin binding of per- and polyfluoroalkyl substances from five structural classes.Toxicol Sci. 2024 Apr 29;199(1):132-149. doi: 10.1093/toxsci/kfae028. Toxicol Sci. 2024. PMID: 38518100 Free PMC article.
-
Role of bioavailability and protein binding of four anionic perfluoroalkyl substances in cell-based bioassays for quantitative in vitro to in vivo extrapolations.Environ Int. 2023 Mar;173:107857. doi: 10.1016/j.envint.2023.107857. Epub 2023 Mar 1. Environ Int. 2023. PMID: 36881956
-
Protein and lipid binding parameters in rainbow trout (Oncorhynchus mykiss) blood and liver fractions to extrapolate from an in vitro metabolic degradation assay to in vivo bioaccumulation potential of hydrophobic organic chemicals.Chem Res Toxicol. 2011 Jul 18;24(7):1134-43. doi: 10.1021/tx200114y. Epub 2011 Jun 8. Chem Res Toxicol. 2011. PMID: 21604782
-
The sources and bioaccumulation of per- and polyfluoroalkyl substances in animal-derived foods and the potential risk of dietary intake.Sci Total Environ. 2023 Dec 20;905:167313. doi: 10.1016/j.scitotenv.2023.167313. Epub 2023 Sep 22. Sci Total Environ. 2023. PMID: 37742961 Review.
-
Associations between Per- and Polyfluoroalkyl Substances Exposures and Blood Lipid Levels among Adults-A Meta-Analysis.Environ Health Perspect. 2023 May;131(5):56001. doi: 10.1289/EHP11840. Epub 2023 May 4. Environ Health Perspect. 2023. PMID: 37141244 Free PMC article. Review.
References
-
- Numata J.; Kowalczyk J.; Adolphs J.; Ehlers S.; Schafft H.; Fuerst P.; Muller-Graf C.; Lahrssen-Wiederholt M.; Greiner M. Toxicokinetics of seven perfluoroalkyl sulfonic and carboxylic acids in pigs fed a contaminated diet. J. Agric. Food Chem. 2014, 62 (28), 6861–6870. 10.1021/jf405827u. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

