A prospective randomized controlled clinical trial investigating the efficacy of low-dose olanzapine in preventing nausea and vomiting associated with oxaliplatin-based and irinotecan-based chemotherapy
- PMID: 38806870
- PMCID: PMC11133208
- DOI: 10.1007/s00432-024-05712-7
A prospective randomized controlled clinical trial investigating the efficacy of low-dose olanzapine in preventing nausea and vomiting associated with oxaliplatin-based and irinotecan-based chemotherapy
Abstract
Objective: The aim of this study is to assess the clinical efficacy of a 5 mg dosage of olanzapine in preventing chemotherapy-induced nausea and vomiting (CINV) associated with moderately emetogenic chemotherapy (MEC) among female patients diagnosed with gastrointestinal tract tumors.
Methods: Patients undergoing the oxaliplatin/irinotecan chemotherapy regimen were enrolled in this prospective controlled study. The olanzapine group received a 5 mg dosage of olanzapine along with palonosetron and dexamethasone, while the control group received a standard two-combination regimen consisting of dexamethasone and palonosetron. The primary endpoints included the total protection (TP) rates for the entire age group and the subgroup aged 60 years and above. Secondary endpoints encompassed the total protection rates during the acute and delayed phases within the two age brackets, as well as the total control (TC) rates and complete remission (CR) rates across all three phases (total, acute, and delayed). Additionally, the study involved the assessment of quality of life and the collection of adverse events associated with the interventions.
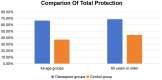
Results: 1) Regarding the primary endpoint, the total phase TP rates within both the entire age group and the age group exceeding 60 years demonstrated superiority in the olanzapine group when compared to the control group (66.7% vs 37.25%, P = 0.003; 68.8% vs 44.4%, P = 0.044). 2) In terms of secondary endpoints, the olanzapine group exhibited superior acute phase TP rates in both age brackets when compared to the control group (P < 0.05). The olanzapine group also demonstrated higher delayed-phase TP rates, TC rates across all three phases, and CR rates within the two age brackets, although the differences were not statistically significant (P > 0.05). Furthermore, the quality of life in the olanzapine group surpassed that of the control group for both age brackets (P < 0.05), characterized by enhanced appetite and a higher incidence of drowsiness in the patients treated with olanzapine when compared to those in the control group (P < 0.05).
Conclusion: Olanzapine can enhance CINV induced by MEC regimen in female patients across all age groups, including the elderly, and therefore improve the quality of life for these patients.
Clinical trial registration: https://www.chictr.org.cn/index.html , identifier: ChiCTR20000368269, 25/08/2020.
Keywords: CINV; Entire age bracket; Female; Low-dose olanzapine; Moderately emetogenic chemotherapy.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Bun S, Yonemori K, Akagi T, Noguchi E, Shimoi T, Shimomura A, Yunokawa M, Shimizu C, Fujiwara Y, Makino Y, Hayashi Y, Tamura K (2018) Feasibility of olanzapine, multi acting receptor targeted antipsychotic agent, for the prevention of emesis caused by continuous cisplatin- or ifosfamide-based chemotherapy. Invest New Drugs 36(1):151–155. 10.1007/s10637-017-0487-3 - DOI - PubMed
-
- Cremolini C, Antoniotti C, Rossini D, Lonardi S, Loupakis F, Pietrantonio F, Bordonaro R, Latiano TP, Tamburini E, Santini D, Passardi A, Marmorino F, Grande R, Aprile G, Zaniboni A, Murgioni S, Granetto C, Buonadonna A, Moretto R, Corallo S, Cordio S, Antonuzzo L, Tomasello G, Masi G, Ronzoni M, Di Donato S, Carlomagno C, Clavarezza M, Ritorto G, Mambrini A, Roselli M, Cupini S, Mammoliti S, Fenocchio E, Corgna E, Zagonel V, Fontanini G, Ugolini C, Boni L, Falcone A, GONO Foundation Investigators (2020) Upfront FOLFOXIRI plus bevacizumab and reintroduction after progression versus mFOLFOX6 plus bevacizumab followed by FOLFIRI plus bevacizumab in the treatment of patients with metastatic colorectal cancer (TRIBE2): a multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 10.1016/S1470-2045(19)30862-9 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical