Jingfang granules protects against intracerebral hemorrhage by inhibiting neuroinflammation and protecting blood-brain barrier damage
- PMID: 38809507
- PMCID: PMC11164481
- DOI: 10.18632/aging.205854
Jingfang granules protects against intracerebral hemorrhage by inhibiting neuroinflammation and protecting blood-brain barrier damage
Abstract
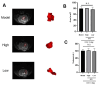
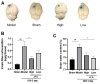
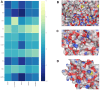
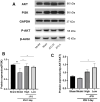
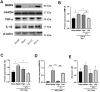
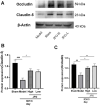
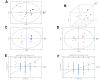
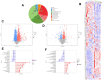
Intracerebral hemorrhage (ICH) can induce intensive oxidative stress, neuroinflammation, and brain cell apoptosis. However, conventional methods for ICH treatment have many disadvantages. There is an urgent need for alternative, effective therapies with minimal side effects. Pharmacodynamics experiment, molecular docking, network pharmacology, and metabolomics were adopted to investigate the treatment and its mechanism of Jingfang Granules (JFG) in ICH. In this study, we investigated the therapeutic effects of JFG on ICH using behavioral, brain water content and Magnetic resonance imaging experiments. However, the key active component and targets of JFG remain unknown. Here we verified that JFG was beneficial to improve brain injury after ICH. A network pharmacology analysis revealed that the anti-inflammatory effect of JFG is predominantly mediated by its activation of the phosphatidylinositol 3-kinase (PI3K)/AKT pathway through Luteolin, (+)-Anomalin and Phaseol and their targeting of AKT1, tumor necrosis factorα (TNF-α), and interleukin-1β (IL-1β). Molecular docking analyses revealed an average affinity of -8.633 kcal/mol, indicating a binding strength of less than -5 kcal/mol. Metabolomic analysis showed that JFG exerted its therapeutic effect on ICH by regulating metabolic pathways, such as the metabolism of taurine and hypotaurine, biosynthesis of valine, leucine, and isoleucine. In conclusion, we demonstrated that JFG attenuated neuroinflammation and BBB injury subsequent to ICH by activating the PI3K/Akt signaling pathway.
Keywords: intracerebral hemorrhage; jingfang granules; metabolomics; network pharmacology; neuroinflammation.
Conflict of interest statement
Figures






References
-
- Wang Y, Jing J, Meng X, Pan Y, Wang Y, Zhao X, Lin J, Li W, Jiang Y, Li Z, Zhang X, Yang X, Ji R, et al. The Third China National Stroke Registry (CNSR-III) for patients with acute ischaemic stroke or transient ischaemic attack: design, rationale and baseline patient characteristics. Stroke Vasc Neurol. 2019; 4:158–64. 10.1136/svn-2019-000242 - DOI - PMC - PubMed
-
- Qi Z. Role and mechanism of IL-23 / IL-17 axis in secondary inflammatory injury after intracerebral hemorrhage, Third Military Medical University. 2017.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

