Streptococcus agalactiae glycolipids promote virulence by thwarting immune cell clearance
- PMID: 38809989
- PMCID: PMC11135403
- DOI: 10.1126/sciadv.adn7848
Streptococcus agalactiae glycolipids promote virulence by thwarting immune cell clearance
Abstract
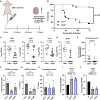
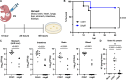
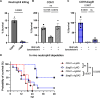
Streptococcus agalactiae [group B Streptococcus (GBS)] is a leading cause of neonatal meningitis, with late-onset disease (LOD) occurring after gastrointestinal tract colonization in infants. Bacterial membrane lipids are essential for host-pathogen interactions, and the functions of glycolipids are yet to be fully elucidated. GBS synthesizes three major glycolipids: glucosyl-diacylglycerol (Glc-DAG), diglucosyl-DAG (Glc2-DAG), and lysyl-Glc-DAG (Lys-Glc-DAG). Here, we identify the enzyme, IagB, as responsible for biosynthesis of Glc-DAG, the precursor for the two other glycolipids in GBS. To examine the collective role of glycolipids to GBS virulence, we adapted a murine model of neonatal meningitis to simulate LOD. The GBS∆iagB mutant traversed the gut-epithelial barrier comparable to wild type but was severely attenuated in bloodstream survival, resulting in decreased bacterial loads in the brain. The GBS∆iagB mutant was more susceptible to neutrophil killing and membrane targeting by host antimicrobial peptides. This work reveals an unexplored function of GBS glycolipids with their ability to protect the bacterial cell from host antimicrobial killing.
Figures




Similar articles
-
Identification of glyoxalase A in group B Streptococcus and its contribution to methylglyoxal tolerance and virulence.Infect Immun. 2025 Apr 8;93(4):e0054024. doi: 10.1128/iai.00540-24. Epub 2025 Feb 26. Infect Immun. 2025. PMID: 40008888 Free PMC article.
-
Identification of a novel cationic glycolipid in Streptococcus agalactiae that contributes to brain entry and meningitis.PLoS Biol. 2022 Feb 18;20(2):e3001555. doi: 10.1371/journal.pbio.3001555. eCollection 2022 Feb. PLoS Biol. 2022. PMID: 35180210 Free PMC article.
-
Virulence and pathogenicity of group B Streptococcus: Virulence factors and their roles in perinatal infection.Virulence. 2025 Dec;16(1):2451173. doi: 10.1080/21505594.2025.2451173. Epub 2025 Jan 23. Virulence. 2025. PMID: 39844743 Free PMC article. Review.
-
Intrapartum antibiotics for known maternal Group B streptococcal colonization.Cochrane Database Syst Rev. 2009 Jul 8;(3):CD007467. doi: 10.1002/14651858.CD007467.pub2. Cochrane Database Syst Rev. 2009. Update in: Cochrane Database Syst Rev. 2013 Jan 31;(1):CD007467. doi: 10.1002/14651858.CD007467.pub3. PMID: 19588432 Updated.
-
Salmonella exploits host- and bacterial-derived β-alanine for replication inside host macrophages.Elife. 2025 Jun 19;13:RP103714. doi: 10.7554/eLife.103714. Elife. 2025. PMID: 40536105 Free PMC article.
Cited by
-
Gut flora-derived succinate exacerbates Allergic Airway Inflammation by promoting protein succinylation.Redox Biol. 2025 May;82:103623. doi: 10.1016/j.redox.2025.103623. Epub 2025 Mar 28. Redox Biol. 2025. PMID: 40174477 Free PMC article.
-
Identification of two glycosyltransferases required for synthesis of membrane glycolipids in Clostridioides difficile.bioRxiv [Preprint]. 2025 Jan 14:2025.01.14.632984. doi: 10.1101/2025.01.14.632984. bioRxiv. 2025. Update in: mBio. 2025 Mar 12;16(3):e0351224. doi: 10.1128/mbio.03512-24. PMID: 39868222 Free PMC article. Updated. Preprint.
-
Identification of two glycosyltransferases required for synthesis of membrane glycolipids in Clostridioides difficile.mBio. 2025 Mar 12;16(3):e0351224. doi: 10.1128/mbio.03512-24. Epub 2025 Feb 18. mBio. 2025. PMID: 39964170 Free PMC article.
-
Identification of Glyoxalase A in Group B Streptococcus and its contribution to methylglyoxal tolerance and virulence.bioRxiv [Preprint]. 2024 Dec 19:2024.07.30.605887. doi: 10.1101/2024.07.30.605887. bioRxiv. 2024. Update in: Infect Immun. 2025 Apr 08;93(4):e0054024. doi: 10.1128/iai.00540-24. PMID: 39131367 Free PMC article. Updated. Preprint.
-
Identification of glyoxalase A in group B Streptococcus and its contribution to methylglyoxal tolerance and virulence.Infect Immun. 2025 Apr 8;93(4):e0054024. doi: 10.1128/iai.00540-24. Epub 2025 Feb 26. Infect Immun. 2025. PMID: 40008888 Free PMC article.
References
-
- E. S. Cowley, I. Z. Chaves, F. Osman, G. Suen, K. Anantharaman, A. J. Hryckowian, Determinants of gastrointestinal group B Streptococcus carriage in adults. bioRxiv 553755 [Preprint] (2023). 10.1101/2023.08.17.553755. - DOI
-
- Wilkinson H. W., Group B streptococcal infection in humans. Annu. Rev. Microbiol. 32, 41–57 (1978). - PubMed
-
- Doran K. S., Nizet V., Molecular pathogenesis of neonatal group B streptococcal infection: No longer in its infancy. Mol. Microbiol. 54, 23–31 (2004). - PubMed
-
- Hall J., Adams N. H., Bartlett L., Seale A. C., Lamagni T., Bianchi-Jassir F., Lawn J. E., Baker C. J., Cutland C., Heath P. T., Ip M., Doare K. L., Madhi S. A., Rubens C. E., Saha S. K., Schrag S., Meulen A. S.-T., Vekemans J., Gravett M. G., Maternal disease with group B streptococcus and serotype distribution worldwide: Systematic review and meta-analyses. Clin. Infect. Dis. 65, S112–S124 (2017). - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

