Interaction of carbonic anhydrase I released from red blood cells with human plasma in vitro
- PMID: 38811147
- PMCID: PMC11188540
- DOI: 10.1093/mtomcs/mfae028
Interaction of carbonic anhydrase I released from red blood cells with human plasma in vitro
Abstract
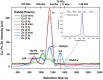
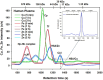
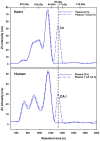
Red blood cells (RBCs) constitute ∼50% of the bloodstream and represent an important target for environmental pollutants and bacterial/viral infections, which can result in their rupture. In addition, diseases such as sickle cell anaemia and paroxysmal nocturnal haemoglobinuria can also result in the rupture of RBCs, which can be potentially life-threatening. With regard to the release of cytosolic metalloproteins from RBCs into the blood-organ system, the biochemical fate of haemoglobin is rather well understood, while comparatively little is known about another highly abundant Zn-metalloprotein, carbonic anhydrase (CA I). To gain insight into the interaction of CA I with human blood plasma constituents, we have employed a metallomics tool comprised of size-exclusion chromatography (SEC) coupled online with an inductively coupled plasma atomic emission spectrometer (ICP-AES), which allows to simultaneously observe all Cu, Fe, and Zn-metalloproteins. After the addition of CA I to human blood plasma incubated at 37°C, the SEC-ICP-AES analysis using phosphate buffered saline (pH 7.4) after 5 min, 1 h, and 2 h revealed that CA I eluted after all endogenous Zn-metalloproteins in the 30 kDa range. Matrix-assisted laser desorption-time of flight mass spectrometry analysis of the collected Zn-peak confirmed that CA I eluted from the column intact. Our in vitro results suggest that CA I released from RBCs to plasma remains free and may be actively involved in health-relevant adverse processes that unfold at the bloodstream-endothelial interface, including atherosclerosis and vision loss.
Keywords: Endothelium; Mechanism of toxicity; Plasma protein binding; Red blood cell lysis; Toxic metals; Zn-metalloprotein.
© The Author(s) 2024. Published by Oxford University Press.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Simultaneous Cu-, Fe-, and Zn-specific detection of metalloproteins contained in rabbit plasma by size-exclusion chromatography-inductively coupled plasma atomic emission spectroscopy.J Biol Inorg Chem. 2009 Jan;14(1):61-74. doi: 10.1007/s00775-008-0424-1. Epub 2008 Sep 10. J Biol Inorg Chem. 2009. PMID: 18781345
-
Removal of Fe3+ and Zn2+ from plasma metalloproteins by iron chelating therapeutics depicted with SEC-ICP-AES.Dalton Trans. 2010 Aug 28;39(32):7466-73. doi: 10.1039/c0dt00229a. Epub 2010 Jul 12. Dalton Trans. 2010. PMID: 20623073
-
Remarkable effect of mobile phase buffer on the SEC-ICP-AES derived Cu, Fe and Zn-metalloproteome pattern of rabbit blood plasma.Metallomics. 2010 Jul;2(7):460-8. doi: 10.1039/c003321a. Epub 2010 May 27. Metallomics. 2010. PMID: 21072345
-
Analysis of the plasma metalloproteome by SEC-ICP-AES: bridging proteomics and metabolomics.Expert Rev Proteomics. 2009 Jun;6(3):251-65. doi: 10.1586/epr.09.44. Expert Rev Proteomics. 2009. PMID: 19489698 Review.
-
Application of a Novel Metallomics Tool to Probe the Fate of Metal-Based Anticancer Drugs in Blood Plasma: Potential, Challenges and Prospects.Curr Top Med Chem. 2021;21(1):48-58. doi: 10.2174/1568026620666200628023540. Curr Top Med Chem. 2021. PMID: 32600232 Review.
Cited by
-
Toward a Mechanism-Driven Integrated Framework to Link Human Exposure to Multiple Toxic Metal(loid) Species with Environmental Diseases.Int J Mol Sci. 2024 Mar 16;25(6):3393. doi: 10.3390/ijms25063393. Int J Mol Sci. 2024. PMID: 38542366 Free PMC article. Review.
References
-
- Maret W., Metallomics. A Primer of Integrated Biometal Sciences. London: Imperial College Press, 2016.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

