Horizontal-cell like Dm9 neurons in Drosophila modulate photoreceptor output to supply multiple functions in early visual processing
- PMID: 38813436
- PMCID: PMC11133737
- DOI: 10.3389/fnmol.2024.1347540
Horizontal-cell like Dm9 neurons in Drosophila modulate photoreceptor output to supply multiple functions in early visual processing
Abstract
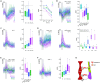
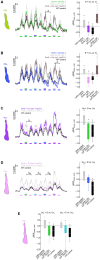
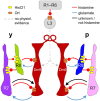
Dm9 neurons in Drosophila have been proposed as functional homologs of horizontal cells in the outer retina of vertebrates. Here we combine genetic dissection of neuronal circuit function, two-photon calcium imaging in Dm9 and inner photoreceptors, and immunohistochemical analysis to reveal novel insights into the functional role of Dm9 in early visual processing. Our experiments show that Dm9 receive input from all four types of inner photoreceptor R7p, R7y, R8p, and R8y. Histamine released from all types R7/R8 directly inhibits Dm9 via the histamine receptor Ort, and outweighs simultaneous histamine-independent excitation of Dm9 by UV-sensitive R7. Dm9 in turn provides inhibitory feedback to all R7/R8, which is sufficient for color-opponent processing in R7 but not R8. Color opponent processing in R8 requires additional synaptic inhibition by R7 of the same ommatidium via axo-axonal synapses and the second Drosophila histamine receptor HisCl1. Notably, optogenetic inhibition of Dm9 prohibits color opponent processing in all types of R7/R8 and decreases intracellular calcium in photoreceptor terminals. The latter likely results from reduced release of excitatory glutamate from Dm9 and shifts overall photoreceptor sensitivity toward higher light intensities. In summary, our results underscore a key role of Dm9 in color opponent processing in Drosophila and suggest a second role of Dm9 in regulating light adaptation in inner photoreceptors. These novel findings on Dm9 are indeed reminiscent of the versatile functions of horizontal cells in the vertebrate retina.
Keywords: color opponency; color vision; feedback inhibition; histamine receptor; horizontal cell; insect; photoreceptor; presynaptic calcium.
Copyright © 2024 Schnaitmann, Pagni, Meyer, Steinhoff, Oberhauser and Reiff.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
Color Processing in the Early Visual System of Drosophila.Cell. 2018 Jan 11;172(1-2):318-330.e18. doi: 10.1016/j.cell.2017.12.018. Cell. 2018. PMID: 29328919
-
Interaction of "chromatic" and "achromatic" circuits in Drosophila color opponent processing.Curr Biol. 2021 Apr 26;31(8):1687-1698.e4. doi: 10.1016/j.cub.2021.01.105. Epub 2021 Feb 25. Curr Biol. 2021. PMID: 33636123
-
Phototactic T-maze Behavioral Assay for Comparing the Functionality of Color-sensitive Photoreceptor Subtypes in the Drosophila Visual System.Bio Protoc. 2020 Mar 20;10(6):e3558. doi: 10.21769/BioProtoc.3558. eCollection 2020 Mar 20. Bio Protoc. 2020. PMID: 33659530 Free PMC article.
-
The development and function of neuronal subtypes processing color and skylight polarization in the optic lobes of Drosophila melanogaster.Arthropod Struct Dev. 2021 Mar;61:101012. doi: 10.1016/j.asd.2020.101012. Epub 2021 Feb 19. Arthropod Struct Dev. 2021. PMID: 33618155 Review.
-
Signaling mechanisms in induction of the R7 photoreceptor in the developing Drosophila retina.Bioessays. 1994 Apr;16(4):237-44. doi: 10.1002/bies.950160406. Bioessays. 1994. PMID: 8031300 Review.
References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

