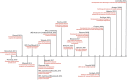
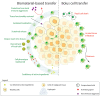
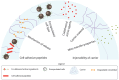
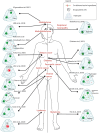
Biomaterial-Based Therapeutic Delivery of Immune Cells
- PMID: 38813869
- PMCID: PMC11607182
- DOI: 10.1002/adhm.202400586
Biomaterial-Based Therapeutic Delivery of Immune Cells
Abstract
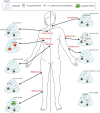
Immune cell therapy (ICT) is a transformative approach used to treat a wide range of diseases including type 1 diabetes, sickle cell disease, disorders of the hematopoietic system, and certain forms of cancers. Despite excellent clinical successes, the scope of adoptively transferred immune cells is limited because of toxicities like cytokine release syndrome and immune effector cell-associated neurotoxicity in patients. Furthermore, reports suggest that such treatment can impact major organ systems including cardiac, renal, pulmonary, and hepatic systems in the long term. Additionally, adoptively transferred immune cells cannot achieve significant penetration into solid tissues, thus limiting their therapeutic potential. Recent studies suggest that biomaterial-assisted delivery of immune cells can address these challenges by reducing toxicity, improving localization, and maintaining desired phenotypes to eventually regain tissue function. In this review, recent efforts in the field of biomaterial-based immune cell delivery for the treatment of diseases, their pros and cons, and where these approaches stand in terms of clinical treatment are highlighted.
Keywords: biomaterials; cancers; cell therapies; diabetes; hydrogels; inflammatory diseases.
© 2024 The Author(s). Advanced Healthcare Materials published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- What's New for Biologics, Latest News from the Center for Biologics Evaluation and Research at FDA, U.S. Food and Drug Administration, [Available from, https://www.fda.gov/vaccines‐blood‐biologics/news‐events‐biologics/whats....
-
- Kamath A., Drug Discovery Today Technol. 2016, 21‐22, 75. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

