Neurexins control the strength and precise timing of glycinergic inhibition in the auditory brainstem
- PMID: 38814174
- PMCID: PMC11139475
- DOI: 10.7554/eLife.94315
Neurexins control the strength and precise timing of glycinergic inhibition in the auditory brainstem
Abstract
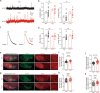
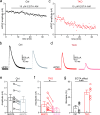
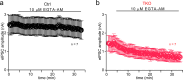
Neurexins play diverse functions as presynaptic organizers in various glutamatergic and GABAergic synapses. However, it remains unknown whether and how neurexins are involved in shaping functional properties of the glycinergic synapses, which mediate prominent inhibition in the brainstem and spinal cord. To address these issues, we examined the role of neurexins in a model glycinergic synapse between the principal neuron in the medial nucleus of the trapezoid body (MNTB) and the principal neuron in the lateral superior olive (LSO) in the auditory brainstem. Combining RNAscope with stereotactic injection of AAV-Cre in the MNTB of neurexin1/2/3 conditional triple knockout mice, we showed that MNTB neurons highly express all isoforms of neurexins although their expression levels vary remarkably. Selective ablation of all neurexins in MNTB neurons not only reduced the amplitude but also altered the kinetics of the glycinergic synaptic transmission at LSO neurons. The synaptic dysfunctions primarily resulted from an impaired Ca2+ sensitivity of release and a loosened coupling between voltage-gated Ca2+ channels and synaptic vesicles. Together, our current findings demonstrate that neurexins are essential in controlling the strength and temporal precision of the glycinergic synapse, which therefore corroborates the role of neurexins as key presynaptic organizers in all major types of fast chemical synapses.
Keywords: LSO; MNTB; auditory; glycinergic; mouse; neurexins; neuroscience; synapse.
© 2024, Jiang et al.
Conflict of interest statement
HJ, RX, XN, ZS, XX, RP, YZ, FL No competing interests declared
Figures






Update of
- doi: 10.1101/2023.12.14.571678
- doi: 10.7554/eLife.94315.1
- doi: 10.7554/eLife.94315.2
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

