Rescue in vitro maturation using ovarian support cells of human oocytes from conventional stimulation cycles yields oocytes with improved nuclear maturation and transcriptomic resemblance to in vivo matured oocytes
- PMID: 38814543
- PMCID: PMC11339229
- DOI: 10.1007/s10815-024-03143-4
Rescue in vitro maturation using ovarian support cells of human oocytes from conventional stimulation cycles yields oocytes with improved nuclear maturation and transcriptomic resemblance to in vivo matured oocytes
Abstract
Purpose: Determine if the gene expression profiles of ovarian support cells (OSCs) and cumulus-free oocytes are bidirectionally influenced by co-culture during in vitro maturation (IVM).
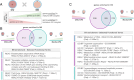
Methods: Fertility patients aged 25 to 45 years old undergoing conventional ovarian stimulation donated denuded immature oocytes for research. Oocytes were randomly allocated to either OSC-IVM culture (intervention) or Media-IVM culture (control) for 24-28 h. The OSC-IVM culture condition was composed of 100,000 OSCs in suspension culture with human chorionic gonadotropin (hCG), recombinant follicle stimulating hormone (rFSH), androstenedione, and doxycycline supplementation. The Media-IVM control lacked OSCs and contained the same supplementation. A limited set of in vivo matured MII oocytes were donated for comparative evaluation. Endpoints consisted of MII formation rate, morphological and spindle quality assessment, and gene expression analysis compared to in vitro and in vivo controls.
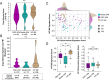
Results: OSC-IVM resulted in a statistically significant improvement in MII formation rate compared to the Media-IVM control, with no apparent effect on morphology or spindle assembly. OSC-IVM MII oocytes displayed a closer transcriptomic maturity signature to IVF-MII controls than Media-IVM control MII oocytes. The gene expression profile of OSCs was modulated in the presence of oocytes, displaying culture- and time-dependent differential gene expression during IVM.
Conclusion: The OSC-IVM platform is a novel tool for rescue maturation of human oocytes, yielding oocytes with improved nuclear maturation and a closer transcriptomic resemblance to in vivo matured oocytes, indicating a potential enhancement in oocyte cytoplasmic maturation. These improvements on oocyte quality after OSC-IVM are possibly occurring through bidirectional crosstalk of cumulus-free oocytes and ovarian support cells.
Keywords: Granulosa cells; In vitro maturation; Oocyte transcriptomics; Ovarian support cells; Stem cells.
© 2024. The Author(s).
Conflict of interest statement
A.D.N., A.B.F., A.G., B.P., C.A., C.C.K., F.B., G.R., K.S.P., K.W., M.M., P.C., S.P. S.K., and M.-J.F.-G. are shareholders in the for-profit biotechnology company Gameto Inc. P.R.J.F. declares paid consultancy for Gameto Inc. P.C. also declares paid consultancy for the Scientific Advisory Board for Gameto Inc. D.H.M. has received consulting services from Granata Bio, Sanford Fertility and Reproductive Medicine, Gameto, and Buffalo IVF, and travel support from the Upper Egypt Assisted Reproduction Society. C.C.K., S.P., M.M., A.G., B.P., K.S.P., G.R., and A.D.N. are listed on a patent covering the use of OSCs for IVM: U.S. Provisional Patent Application No. 63/492,210. Additionally, C.C.K. and K.W. are listed on three patents covering the use of OSCs for IVM: U.S. Patent Application No. 17/846,725, U.S Patent Application No. 17/846,845, and International Patent Application No.: PCT/ US2023/026012. C.C.K., M.P.S., and P.C. additionally are listed on three patents for the transcription factor-directed production of granulosa-like cells from stem cells: International Patent Application No.: PCT/US2023/065140, U.S. Provisional Application No. 63/ 326,640, and U.S. Provisional Application No. 63/444,108. The remaining authors have no conflicts of interest to declare.
Figures


Similar articles
-
Human-induced pluripotent stem cell-derived ovarian support cell co-culture improves oocyte maturation in vitro after abbreviated gonadotropin stimulation.Hum Reprod. 2023 Dec 4;38(12):2456-2469. doi: 10.1093/humrep/dead205. Hum Reprod. 2023. PMID: 37815487 Free PMC article.
-
Pro-cumulin addition in a biphasic in vitro oocyte maturation system modulates human oocyte and cumulus cell transcriptomes.Mol Hum Reprod. 2025 Jan 17;31(1):gaaf001. doi: 10.1093/molehr/gaaf001. Mol Hum Reprod. 2025. PMID: 39862403 Free PMC article.
-
A pre-in vitro maturation medium containing cumulus oocyte complex ligand-receptor signaling molecules maintains meiotic arrest, supports the cumulus oocyte complex and improves oocyte developmental competence.Mol Hum Reprod. 2017 Sep 1;23(9):594-606. doi: 10.1093/molehr/gax032. Mol Hum Reprod. 2017. PMID: 28586460
-
To rescue or not to rescue immature oocytes: prospects and challenges.Fertil Steril. 2025 May;123(5):749-758. doi: 10.1016/j.fertnstert.2025.02.039. Epub 2025 Mar 8. Fertil Steril. 2025. PMID: 40058555 Review.
-
Transcriptomic profiles reveal the characteristics of oocytes and cumulus cells at GV, MI, and MII in follicles before ovulation.J Ovarian Res. 2023 Nov 22;16(1):225. doi: 10.1186/s13048-023-01291-2. J Ovarian Res. 2023. PMID: 37993893 Free PMC article. Review.
Cited by
-
Antioxidant-supplemented media modulates ROS by regulating complex I during mouse oocyte maturation.Sci Rep. 2025 Jul 2;15(1):23029. doi: 10.1038/s41598-025-08056-5. Sci Rep. 2025. PMID: 40596396 Free PMC article.
-
Current status and hotspots of in vitro oocyte maturation: a bibliometric study of the past two decades.J Assist Reprod Genet. 2025 Feb;42(2):459-472. doi: 10.1007/s10815-024-03272-w. Epub 2024 Sep 25. J Assist Reprod Genet. 2025. PMID: 39317914 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

