Chronic intracranial recordings in the globus pallidus reveal circadian rhythms in Parkinson's disease
- PMID: 38816390
- PMCID: PMC11139908
- DOI: 10.1038/s41467-024-48732-0
Chronic intracranial recordings in the globus pallidus reveal circadian rhythms in Parkinson's disease
Abstract
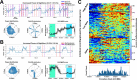
Circadian rhythms have been shown in the subthalamic nucleus (STN) in Parkinson's disease (PD), but only a few studies have focused on the globus pallidus internus (GPi). This retrospective study investigates GPi circadian rhythms in a large cohort of subjects with PD (130 recordings from 93 subjects) with GPi activity chronically recorded in their home environment. We found a significant change in GPi activity between daytime and nighttime in most subjects (82.4%), with a reduction in GPi activity at nighttime in 56.2% of recordings and an increase in activity in 26.2%. GPi activity in higher frequency bands ( > 20 Hz) was more likely to decrease at night and in patients taking extended-release levodopa medication. Our results suggest that circadian fluctuations in the GPi vary across individuals and that increased power at night might be due to the reemergence of pathological neural activity. These findings should be considered to ensure successful implementation of adaptive neurostimulation paradigms in the real-world.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Parkinsonian Beta Dynamics during Rest and Movement in the Dorsal Pallidum and Subthalamic Nucleus.J Neurosci. 2020 Apr 1;40(14):2859-2867. doi: 10.1523/JNEUROSCI.2113-19.2020. Epub 2020 Feb 27. J Neurosci. 2020. PMID: 32107277 Free PMC article.
-
Meta-analysis comparing deep brain stimulation of the globus pallidus and subthalamic nucleus to treat advanced Parkinson disease.J Neurosurg. 2014 Sep;121(3):709-18. doi: 10.3171/2014.4.JNS131711. Epub 2014 Jun 6. J Neurosurg. 2014. PMID: 24905564
-
Pallidal vs subthalamic nucleus deep brain stimulation in Parkinson disease.Arch Neurol. 2005 Apr;62(4):554-60. doi: 10.1001/archneur.62.4.554. Arch Neurol. 2005. PMID: 15824252 Clinical Trial.
-
Congress of Neurological Surgeons Systematic Review and Evidence-Based Guideline on Subthalamic Nucleus and Globus Pallidus Internus Deep Brain Stimulation for the Treatment of Patients With Parkinson's Disease: Executive Summary.Neurosurgery. 2018 Jun 1;82(6):753-756. doi: 10.1093/neuros/nyy037. Neurosurgery. 2018. PMID: 29538685 Free PMC article.
-
Cognition and Depression Following Deep Brain Stimulation of the Subthalamic Nucleus and Globus Pallidus Pars Internus in Parkinson's Disease: A Meta-Analysis.Neuropsychol Rev. 2015 Dec;25(4):439-54. doi: 10.1007/s11065-015-9302-0. Epub 2015 Oct 12. Neuropsychol Rev. 2015. PMID: 26459361 Review.
Cited by
-
KNN-based frequency-adjustable ferroelectric heterojunction and biomedical applications.Nat Commun. 2025 Aug 2;16(1):7120. doi: 10.1038/s41467-025-62079-0. Nat Commun. 2025. PMID: 40753174 Free PMC article.
-
Finely Tuned γ Tracks Medication Cycles in Parkinson's Disease: An Ambulatory Brain-Sense Study.Mov Disord. 2025 May;40(5):881-895. doi: 10.1002/mds.30160. Epub 2025 Mar 13. Mov Disord. 2025. PMID: 40079832 Free PMC article.
-
At-Home Movement State Classification Using Totally Implantable Bidirectional Cortical-Basal Ganglia Neural Interface.Res Sq [Preprint]. 2025 Mar 12:rs.3.rs-6058394. doi: 10.21203/rs.3.rs-6058394/v1. Res Sq. 2025. PMID: 40162212 Free PMC article. Preprint.
-
Stimulation-Evoked Resonant Neural Activity in the Subthalamic Nucleus Is Modulated by Sleep.Mov Disord. 2025 Feb;40(2):351-356. doi: 10.1002/mds.30063. Epub 2024 Nov 19. Mov Disord. 2025. PMID: 39560163 Free PMC article.
-
Tailoring deep brain stimulation for sleep: using actigraphy to understand the relationship between Parkinsonian brain activity and behavioral state.Sleep. 2025 Jun 13;48(6):zsaf066. doi: 10.1093/sleep/zsaf066. Sleep. 2025. PMID: 40088453 Free PMC article. No abstract available.

