Harnessing Hyperthermia: Molecular, Cellular, and Immunological Insights for Enhanced Anticancer Therapies
- PMID: 38818970
- PMCID: PMC11143831
- DOI: 10.1177/15347354241242094
Harnessing Hyperthermia: Molecular, Cellular, and Immunological Insights for Enhanced Anticancer Therapies
Abstract
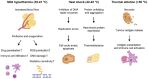
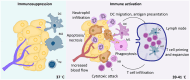
Hyperthermia, the raising of tumor temperature (≥39°C), holds great promise as an adjuvant treatment for cancer therapy. This review focuses on 2 key aspects of hyperthermia: its molecular and cellular effects and its impact on the immune system. Hyperthermia has profound effects on critical biological processes. Increased temperatures inhibit DNA repair enzymes, making cancer cells more sensitive to chemotherapy and radiation. Elevated temperatures also induce cell cycle arrest and trigger apoptotic pathways. Furthermore, hyperthermia modifies the expression of heat shock proteins, which play vital roles in cancer therapy, including enhancing immune responses. Hyperthermic treatments also have a significant impact on the body's immune response against tumors, potentially improving the efficacy of immune checkpoint inhibitors. Mild systemic hyperthermia (39°C-41°C) mimics fever, activating immune cells and raising metabolic rates. Intense heat above 50°C can release tumor antigens, enhancing immune reactions. Using photothermal nanoparticles for targeted heating and drug delivery can also modulate the immune response. Hyperthermia emerges as a cost-effective and well-tolerated adjuvant therapy when integrated with immunotherapy. This comprehensive review serves as a valuable resource for the selection of patient-specific treatments and the guidance of future experimental studies.
Keywords: DNA repair; apoptosis; fever; heat shock protein; immune system; nanoparticle; thermal ablation.
Conflict of interest statement
Declaration of Conflicting InterestsThe author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DECLARATION OF GENERATIVE AI AND AI-ASSISTED TECHNOLOGIES IN THE WRITING PROCESS: During the preparation of this work the authors used ChatGPT to improve grammar. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Figures
Similar articles
-
The role of hyperthermia in the treatment of tumor.Crit Rev Oncol Hematol. 2024 Dec;204:104541. doi: 10.1016/j.critrevonc.2024.104541. Epub 2024 Oct 24. Crit Rev Oncol Hematol. 2024. PMID: 39461607 Review.
-
From cold to hot: mechanisms of hyperthermia in modulating tumor immunology for enhanced immunotherapy.Front Immunol. 2025 Feb 28;16:1487296. doi: 10.3389/fimmu.2025.1487296. eCollection 2025. Front Immunol. 2025. PMID: 40092992 Free PMC article. Review.
-
Combinatorial immunotherapy and nanoparticle mediated hyperthermia.Adv Drug Deliv Rev. 2017 May 15;114:175-183. doi: 10.1016/j.addr.2017.06.008. Epub 2017 Jun 15. Adv Drug Deliv Rev. 2017. PMID: 28625829 Review.
-
Immunogenic Effect of Hyperthermia on Enhancing Radiotherapeutic Efficacy.Int J Mol Sci. 2018 Sep 17;19(9):2795. doi: 10.3390/ijms19092795. Int J Mol Sci. 2018. PMID: 30227629 Free PMC article. Review.
-
Emerging hyperthermia applications for pediatric oncology.Adv Drug Deliv Rev. 2020;163-164:157-167. doi: 10.1016/j.addr.2020.10.016. Epub 2020 Oct 24. Adv Drug Deliv Rev. 2020. PMID: 33203538 Review.
Cited by
-
Hyperthermia and targeting heat shock proteins: innovative approaches for neurodegenerative disorders and Long COVID.Front Neurosci. 2025 Feb 4;19:1475376. doi: 10.3389/fnins.2025.1475376. eCollection 2025. Front Neurosci. 2025. PMID: 39967803 Free PMC article. Review.
-
Harnessing Hyperthermia: Molecular, Cellular, and Immunological Insights for Enhanced Anticancer Therapies: Comment.Integr Cancer Ther. 2024 Jan-Dec;23:15347354241275038. doi: 10.1177/15347354241275038. Integr Cancer Ther. 2024. PMID: 39223848 Free PMC article. No abstract available.
-
Water-filtered infrared A radiation hyperthermia combined with immunotherapy for advanced gastrointestinal tumours.Cancer Med. 2024 Jul;13(14):e70024. doi: 10.1002/cam4.70024. Cancer Med. 2024. PMID: 39049187 Free PMC article.
-
Preliminary Insights into the Acute Molecular Responses in C2C12 Myotubes to Hyperthermia and Insulin Treatment.bioRxiv [Preprint]. 2025 Mar 28:2025.03.26.644592. doi: 10.1101/2025.03.26.644592. bioRxiv. 2025. PMID: 40196696 Free PMC article. Preprint.
-
HSP47 at the Crossroads of Thrombosis and Collagen Dynamics: Unlocking Therapeutic Horizons and Debates.TH Open. 2025 Jun 5;9:a25994925. doi: 10.1055/a-2599-4925. eCollection 2025. TH Open. 2025. PMID: 40486904 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

