Roles of Ferric Peroxide Anion Intermediates (Fe3+O2 -, Compound 0) in Cytochrome P450 19A1 Steroid Aromatization and a Cytochrome P450 2B4 Secosteroid Oxidation Model
- PMID: 38820076
- PMCID: PMC11519728
- DOI: 10.1002/anie.202406542
Roles of Ferric Peroxide Anion Intermediates (Fe3+O2 -, Compound 0) in Cytochrome P450 19A1 Steroid Aromatization and a Cytochrome P450 2B4 Secosteroid Oxidation Model
Abstract
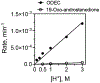
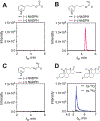
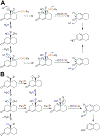
Cytochrome P450 (P450, CYP) 19A1 is the steroid aromatase, the enzyme responsible for the 3-step conversion of androgens (androstenedione or testosterone) to estrogens. The final step is C-C bond scission (removing the 19-oxo group as formic acid) that proceeds via a historically controversial reaction mechanism. The two competing mechanistic possibilities involve a ferric peroxide anion (Fe3+O2 -, Compound 0) and a perferryl oxy species (FeO3+, Compound I). One approach to discern the role of each species in the reaction is with the use of oxygen-18 labeling, i.e., from 18O2 and H2 18O of the reaction product formic acid. We applied this approach, using several technical improvements, to study the deformylation of 19-oxo-androstenedione by human P450 19A1 and of a model secosteroid, 3-oxodecaline-4-ene-10-carboxaldehyde (ODEC), by rabbit P450 2B4. Both aldehyde substrates were sensitive to non-enzymatic acid-catalyzed deformylation, yielding 19-norsteroids, and conditions were established to avoid issues with artifactual generation of formic acid. The Compound 0 reaction pathway predominated (i.e., Fe3+O2 -) in both P450 19A1 oxidation of 19-oxo-androstenedione and P450 2B4 oxidation of ODEC. The P450 19A1 results contrast with our prior conclusions (J. Am. Chem. Soc. 2014, 136, 15016-16025), attributed to several technical modifications.
Keywords: cytochrome P450; enzyme mechanisms; mass spectrometry; oxidation mechanisms; steroid biosynthesis.
© 2024 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Figures


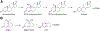
Similar articles
-
Updates on Mechanisms of Cytochrome P450 Catalysis of Complex Steroid Oxidations.Int J Mol Sci. 2024 Aug 20;25(16):9020. doi: 10.3390/ijms25169020. Int J Mol Sci. 2024. PMID: 39201706 Free PMC article. Review.
-
Aromatization of a bicyclic steroid analog, 3-oxodecalin-4-ene-10-carboxaldehyde, by liver microsomal cytochrome P450 2B4.Biochemistry. 1994 Nov 22;33(46):13651-61. doi: 10.1021/bi00250a015. Biochemistry. 1994. PMID: 7947773
-
Mechanism of the third oxidative step in the conversion of androgens to estrogens by cytochrome P450 19A1 steroid aromatase.J Am Chem Soc. 2014 Oct 22;136(42):15016-25. doi: 10.1021/ja508185d. Epub 2014 Oct 10. J Am Chem Soc. 2014. PMID: 25252141 Free PMC article.
-
Kinetic analysis of the three-step steroid aromatase reaction of human cytochrome P450 19A1.J Biol Chem. 2010 Jun 4;285(23):17734-43. doi: 10.1074/jbc.M110.123711. Epub 2010 Apr 12. J Biol Chem. 2010. PMID: 20385561 Free PMC article.
-
Studies on the mechanism of aromatase and other cytochrome P450 mediated deformylation reactions.J Steroid Biochem Mol Biol. 1993 Mar;44(4-6):367-73. doi: 10.1016/0960-0760(93)90240-w. J Steroid Biochem Mol Biol. 1993. PMID: 8476750 Review.
Cited by
-
Updates on Mechanisms of Cytochrome P450 Catalysis of Complex Steroid Oxidations.Int J Mol Sci. 2024 Aug 20;25(16):9020. doi: 10.3390/ijms25169020. Int J Mol Sci. 2024. PMID: 39201706 Free PMC article. Review.
-
Cytochrome P450BM-3 and P450 11A1 retain Compound I (FeO3+) chemistry with electrophilic substrates poised for Compound 0 (Fe3+O2-) reactions.J Biol Chem. 2025 Jul;301(7):110378. doi: 10.1016/j.jbc.2025.110378. Epub 2025 Jun 14. J Biol Chem. 2025. PMID: 40523616 Free PMC article.
-
Dioxygenase Chemistry in Nucleophilic Aldehyde Deformylations Utilizing Dicopper O2-Derived Peroxide Complexes.J Am Chem Soc. 2024 Aug 28;146(34):23854-23871. doi: 10.1021/jacs.4c06243. Epub 2024 Aug 14. J Am Chem Soc. 2024. PMID: 39141923 Free PMC article.
-
Mass Spectral Analysis of Sterols and Other Steroids in Different Ionization Modes: Sensitivity and Oxidation Artifacts.J Am Soc Mass Spectrom. 2025 Aug 6;36(8):1702-1717. doi: 10.1021/jasms.5c00099. Epub 2025 Jun 26. J Am Soc Mass Spectrom. 2025. PMID: 40574312 Free PMC article.
-
Multifunctional cytochrome P450 orchestrates radical cleavage and non-radical cyclization in 5-oxaindolizidine biosynthesis.Chem Sci. 2025 May 10;16(23):10563-10571. doi: 10.1039/d4sc07174c. eCollection 2025 Jun 11. Chem Sci. 2025. PMID: 40375862 Free PMC article.
References
-
- Ortiz de Montellano PR, Cytochrome P450: Structure, Mechanism, and Biochemistry, 4th ed., Springer, New York, 2015;
- Auchus RJ, Miller WL, in Cytochrome P450: Structure, Mechanism, and Biochemistry, Vol. 2, 4th ed. (Ed.: Ortiz de Montellano PR), Springer, New York, 2015, pp. 851–879.
-
- Ortiz de Montellano PR, in Cytochrome P450: Structure, Mechanism, and Biochemistry, 4th ed. (Ed.: Ortiz de Montellano PR), Springer, New York, 2015, pp. 111–176;
- Guengerich FP, Yoshimoto FK, Chem. Rev 2018, 118, 6573–6655; - PMC - PubMed
- Guengerich FP, Tateishi Y, McCarty KD, Med. Chem. Res 2023, 32, 1263–1277.
-
- Hosoda H, Fishman J, J. Am. Chem. Soc 1974, 96, 7325–7329; - PubMed
- Goto J, Fishman J, Science 1977, 195, 80–81; - PubMed
- Hahn EF, Fishman J, J. Biol. Chem 1984, 259, 1689–1694; - PubMed
- Fishman J, Cancer Res 1982, 42, 3277s–3280s; - PubMed
- Townsley JD, Brodie HJ, Biochemistry 1968, 7, 33–40; - PubMed
- Morand P, Williamson DG, Layne DS, Lompa-Krzymien L, Salvador J, Biochemistry 1975, 14, 635–638; - PubMed
- Cole PA, Robinson CH, J. Med. Chem 1990, 33, 2933–2942; - PubMed
- Caspi E, Njar VC, Steroids 1987, 50, 347–362; - PubMed
- Korzekwa KR, Trager WF, Mancewicz J, Osawa Y, Steroid Biochem J. Mol. Biol 1993, 44, 367–373. - PubMed
-
- Caspi E, Wicha J, Arunachalam T, Nelson P, Spiteller G, J. Am. Chem. Soc 1984, 106, 7282–7283;
- Covey DF, Hood WF, Beusen DD, Carrell HL, Biochemistry 1984, 23, 5398–5406; - PubMed
- Ortiz de Montellano PR, in Cytochrome P-450, 1st ed., (Ed.: Ortiz de Montellano PR), Plenum Press, New York, 1986, pp. 217–271.
-
- Cole PA, Robinson CH, J. Am. Chem. Soc 1988, 110, 1284–1285;
- Watanabe Y, Ishimura Y, J. Am. Chem. Soc 1989, 111, 8047–8049;
- Cole PA, Robinson CH, J. Chem. Soc., Perk. Trans 1 1990, 2119–2125;
- Wertz DL, Sisemore MF, Selke M, Driscoll J, Valentine JS, J. Am. Chem. Soc 1998, 120, 5331–5332.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

