Outcomes after surgery for infective endocarditis
- PMID: 38827557
- PMCID: PMC11139833
- DOI: 10.1007/s12055-023-01647-9
Outcomes after surgery for infective endocarditis
Abstract
The role of surgery in infective endocarditis is becoming established the world over. In spite of all recent advances, endocarditis remains a lethal disease following surgery. With the emergence of more difficult-to-treat microorganisms, sicker and older patients with multiple co-morbidities, and an increase in healthcare-associated infections, the need for surgery in the management of infective endocarditis is only bound to increase. Data on the use of surgery in endocarditis till date is largely from observational data due to the relative rarity of the disease and variable practice patterns around the world. Hopefully, with increasing awareness and more inter-institutional and international collaborations, more robust data will emerge to further establish the role of surgery. For the time being, individual patient management will require the active multi-disciplinary approach of an endocarditis team to provide the best possible outcomes.
Supplementary information: The online version contains supplementary material available at 10.1007/s12055-023-01647-9.
Keywords: Endocarditis; Infective endocarditis; Outcome; Surgery.
© Indian Association of Cardiovascular-Thoracic Surgeons 2023. Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Conflict of interest statement
Conflict of interestThe authors declare no conflict of interest.
Figures
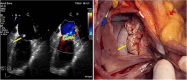
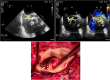
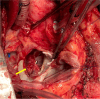
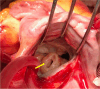
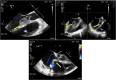
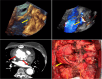
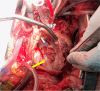
References
-
- Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP III, Gentile F, et al. ACC/AHA guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines [published correction appears in Circulation. 2021 Feb 2;143: e228] [published correction appears in Circulation. 2021 Mar 9;143: e784]. Circulation. 2021;143:e35-e71. 10.1161/CIR.0000000000000932. - PubMed
-
- Chu VH, Park LP, Athan E, Delahaye F, Freiberger T, Lamas C, et al. Association between surgical indications, operative risk, and clinical outcome in infective endocarditis: a prospective study from the International Collaboration on Endocarditis. Circulation. 2015;131:131–140. doi: 10.1161/CIRCULATIONAHA.114.012461. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
