Hearing loss in Australian First Nations children at 6-monthly assessments from age 12 to 36 months: Secondary outcomes from randomised controlled trials of novel pneumococcal conjugate vaccine schedules
- PMID: 38829821
- PMCID: PMC11146696
- DOI: 10.1371/journal.pmed.1004375
Hearing loss in Australian First Nations children at 6-monthly assessments from age 12 to 36 months: Secondary outcomes from randomised controlled trials of novel pneumococcal conjugate vaccine schedules
Abstract
Background: In Australian remote communities, First Nations children with otitis media (OM)-related hearing loss are disproportionately at risk of developmental delay and poor school performance, compared to those with normal hearing. Our objective was to compare OM-related hearing loss in children randomised to one of 2 pneumococcal conjugate vaccine (PCV) formulations.
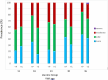
Methods and findings: In 2 sequential parallel, open-label, randomised controlled trials (the PREVIX trials), eligible infants were first allocated 1:1:1 at age 28 to 38 days to standard or mixed PCV schedules, then at age 12 months to PCV13 (13-valent pneumococcal conjugate vaccine, +P) or PHiD-CV10 (10-valent pneumococcal Haemophilus influenzae protein D conjugate vaccine, +S) (1:1). Here, we report prevalence and level of hearing loss outcomes in the +P and +S groups at 6-monthly scheduled assessments from age 12 to 36 months. From March 2013 to September 2018, 261 infants were enrolled and 461 hearing assessments were performed. Prevalence of hearing loss was 78% (25/32) in the +P group and 71% (20/28) in the +S group at baseline, declining to 52% (28/54) in the +P groups and 56% (33/59) in the +S group at age 36 months. At primary endpoint age 18 months, prevalence of moderate (disabling) hearing loss was 21% (9/42) in the +P group and 41% (20/49) in the +S group (difference -19%; (95% confidence interval (CI) [-38, -1], p = 0.07) and prevalence of no hearing loss was 36% (15/42) in the +P group and 16% (8/49) in the +S group (difference 19%; (95% CI [2, 37], p = 0.05). At subsequent time points, prevalence of moderate hearing loss remained lower in the +P group: differences -3%; (95% CI [-23, 18], p = 1.00 at age 24 months), -12%; (95% CI [-30, 6], p = 0.29 at age 30 months), and -9%; (95% CI [-23, 5], p = 0.25 at age 36 months). A major limitation was the small sample size, hence low power to reach statistical significance, thereby reducing confidence in the effect size.
Conclusions: In this study, we observed a high prevalence and persistence of moderate (disabling) hearing loss throughout early childhood. We found a lower prevalence of moderate hearing loss and correspondingly higher prevalence of no hearing loss in the +P group, which may have substantial benefits for high-risk children, their families, and society, but warrant further investigation.
Trial registration: ClinicalTrials.gov NCT01735084 and NCT01174849.
Copyright: © 2024 Leach et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
AJL received funds from NHMRC paid to the institution, and GSK provided materials for immunogenicity assays. AJL received funds from Merck Sharp and Dohme for analysis of pneumococcal carriage, payment to institution. ABC served as advisor on a Data Safety Monitoring Board for an unlicensed vaccine (GlaxoSmithKline) and an unlicensed monoclonal antibody (AstraZeneca), was an adviser on an unlicensed molecule for chronic cough (Merck); and has multiple project grants and a Centre of Research Excellence relating to various aspects of bronchiectasis in children from the National Health and Medical Research Council. ABC received Royalties or licences as an author of cough and bronchiectasis topics, and Partial reimbursement for airfares as a speaker for European Respiratory Society. All payments were to the institution. PM served on a data safety and monitoring board for the Novavax COVID-19 vaccine. JB provided a report to Merck Sharp and Dohme Australia. All other authors (DL, CGB-J, HS-V, MJB, MDC, PVL, PSM, PJT) declare no competing interests.
Figures

References
-
- Su JY, Guthridge S, He VY, Howard D, Leach AJ. The impact of hearing impairment on early academic achievement in Aboriginal children living in remote Australia: a data linkage study. BMC Public Health. 2020;20(1):1521. Epub 2020/10/09. doi: 10.1186/s12889-020-09620-6 ; PubMed Central PMCID: PMC33028291. - DOI - PMC - PubMed
-
- Leach AJ, Wigger C, Hare K, Hampton V, Beissbarth J, Andrews R, et al.. Reduced middle ear infection with non-typeable Haemophilus influenzae, but not Streptococcus pneumoniae, after transition to 10-valent pneumococcal non-typeable H. influenzae protein D conjugate vaccine. BMC Pediatr. 2015;15(1):162–175. Epub 2015/10/21. doi: 10.1186/s12887-015-0483-8 ; PubMed Central PMCID: PMC4615539. - DOI - PMC - PubMed
-
- Leach AJ, Wigger C, Beissbarth J, Woltring D, Andrews R, Chatfield MD, et al.. General health, otitis media, nasopharyngeal carriage and middle ear microbiology in Northern Territory Aboriginal children vaccinated during consecutive periods of 10-valent or 13-valent pneumococcal conjugate vaccines. Int J Pediatr Otorhinolaryngol. 2016;86:224–232. Epub 2016/06/05. doi: 10.1016/j.ijporl.2016.05.011 . - DOI - PubMed
-
- Leach AJ, Mulholland EK, Santosham M, Torzillo PJ, McIntyre P, Smith-Vaughan H, et al.. Interchangeability, immunogenicity and safety of a combined 10-valent pneumococcal Haemophilus influenzae protein D conjugate vaccine (Synflorix) and 13-valent-PCV (Prevenar13) schedule at 1-2-4-6 months: PREVIX_COMBO, a 3-arm randomised controlled trial. Vaccine X. 2021;7:100086. doi: 10.1016/j.jvacx.2021.100086 - DOI - PMC - PubMed
-
- Leach AJ, Mulholland EK, Santosham M, Torzillo PJ, Brown NJ, McIntyre P, et al.. Pneumococcal conjugate vaccines PREVenar13 and SynflorIX in sequence or alone in high-risk Indigenous infants (PREV-IX_COMBO): protocol of a randomised controlled trial. BMJ Open. 2015;5(1):e007247–e007257. Epub 2015/01/18. doi: 10.1136/bmjopen-2014-007247 . - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

