Elucidating the spatio-temporal dynamics of the Plasmodium falciparum basal complex
- PMID: 38829893
- PMCID: PMC11175456
- DOI: 10.1371/journal.ppat.1012265
Elucidating the spatio-temporal dynamics of the Plasmodium falciparum basal complex
Abstract
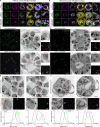
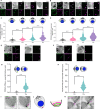
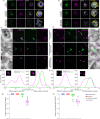
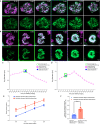
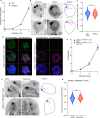
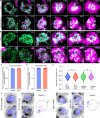
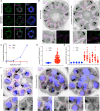
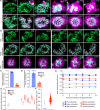
Asexual replication of Plasmodium falciparum occurs via schizogony, wherein 16-36 daughter cells are produced within the parasite during one semi-synchronized cytokinetic event. Schizogony requires a divergent contractile ring structure known as the basal complex. Our lab has previously identified PfMyoJ (PF3D7_1229800) and PfSLACR (PF3D7_0214700) as basal complex proteins recruited midway through segmentation. Using ultrastructure expansion microscopy, we localized both proteins to a novel basal complex subcompartment. While both colocalize with the basal complex protein PfCINCH upon recruitment, they form a separate, more basal subcompartment termed the posterior cup during contraction. We also show that PfSLACR is recruited to the basal complex prior to PfMyoJ, and that both proteins are removed unevenly as segmentation concludes. Using live-cell microscopy, we show that actin dynamics are dispensable for basal complex formation, expansion, and contraction. We then show that EF-hand containing P. falciparum Centrin 2 partially localizes to this posterior cup of the basal complex and that it is essential for growth and replication, with variable defects in basal complex contraction and synchrony. Finally, we demonstrate that free intracellular calcium is necessary but not sufficient for basal complex contraction in P. falciparum. Thus, we demonstrate dynamic spatial compartmentalization of the Plasmodium falciparum basal complex, identify an additional basal complex protein, and begin to elucidate the unique mechanism of contraction utilized by P. falciparum, opening the door for further exploration of Apicomplexan cellular division.
Copyright: © 2024 Morano et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- World Health Organization. World Malaria Report 2021. 2021.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

