Developmental hearing loss-induced perceptual deficits are rescued by genetic restoration of cortical inhibition
- PMID: 38830095
- PMCID: PMC11181144
- DOI: 10.1073/pnas.2311570121
Developmental hearing loss-induced perceptual deficits are rescued by genetic restoration of cortical inhibition
Abstract
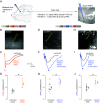
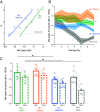
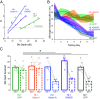
Even a transient period of hearing loss during the developmental critical period can induce long-lasting deficits in temporal and spectral perception. These perceptual deficits correlate with speech perception in humans. In gerbils, these hearing loss-induced perceptual deficits are correlated with a reduction of both ionotropic GABAA and metabotropic GABAB receptor-mediated synaptic inhibition in auditory cortex, but most research on critical period plasticity has focused on GABAA receptors. Therefore, we developed viral vectors to express proteins that would upregulate gerbil postsynaptic inhibitory receptor subunits (GABAA, Gabra1; GABAB, Gabbr1b) in pyramidal neurons, and an enzyme that mediates GABA synthesis (GAD65) presynaptically in parvalbumin-expressing interneurons. A transient period of developmental hearing loss during the auditory critical period significantly impaired perceptual performance on two auditory tasks: amplitude modulation depth detection and spectral modulation depth detection. We then tested the capacity of each vector to restore perceptual performance on these auditory tasks. While both GABA receptor vectors increased the amplitude of cortical inhibitory postsynaptic potentials, only viral expression of postsynaptic GABAB receptors improved perceptual thresholds to control levels. Similarly, presynaptic GAD65 expression improved perceptual performance on spectral modulation detection. These findings suggest that recovering performance on auditory perceptual tasks depends on GABAB receptor-dependent transmission at the auditory cortex parvalbumin to pyramidal synapse and point to potential therapeutic targets for developmental sensory disorders.
Keywords: auditory cortex; critical period; hearing loss; inhibition; parvalbumin.
Conflict of interest statement
Competing interests statement:S.M. and D.H.S. submitted a patent application for the viruses used to express Gabra1 and Gabbr1b (U.S. Patent Application No. 63/359,724), and a provisional patent application for the virus to express GAD65 (U.S. Patent Provisional Application No. 63/507,894). R.F. and T.M.M. declare no competing financial interests.
Figures

Update of
-
Developmental hearing loss-induced perceptual deficits are rescued by cortical expression of GABAB receptors.bioRxiv [Preprint]. 2023 Jan 10:2023.01.10.523440. doi: 10.1101/2023.01.10.523440. bioRxiv. 2023. Update in: Proc Natl Acad Sci U S A. 2024 Jun 11;121(24):e2311570121. doi: 10.1073/pnas.2311570121. PMID: 36711464 Free PMC article. Updated. Preprint.
References
-
- Braat S., Kooy R. F., The GABAA receptor as a therapeutic target for neurodevelopmental disorders. Neuron 86, 1119–1130 (2015). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

