Proton-driven sodium secretion in a saline water animal
- PMID: 38830894
- PMCID: PMC11148202
- DOI: 10.1038/s41598-024-62974-4
Proton-driven sodium secretion in a saline water animal
Erratum in
-
Author Correction: Proton-driven sodium secretion in a saline water animal.Sci Rep. 2024 Oct 10;14(1):23706. doi: 10.1038/s41598-024-74220-y. Sci Rep. 2024. PMID: 39390110 Free PMC article. No abstract available.
Abstract
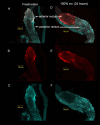
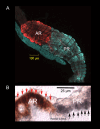
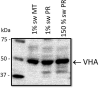
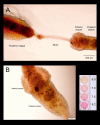
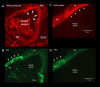
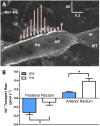
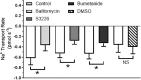
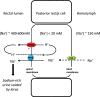
Aquatic animals residing in saline habitats either allow extracellular sodium concentration to conform to environmental values or regulate sodium to lower levels. The latter strategy requires an energy-driven process to move sodium against a large concentration gradient to eliminate excess sodium that diffuses into the animal. Previous studies of invertebrate and vertebrate species indicate a sodium pump, Na+/K+ ATPase, powers sodium secretion. We provide the first functional evidence of a saline-water animal, Aedes taeniorhynchus mosquito larva, utilizing a proton pump to power this process. Vacuolar-type H+ ATPase (VHA) protein is highly expressed on the apical membrane of the posterior rectal cells, and in situ sodium flux across this epithelium increases significantly in larvae held in higher salinity and is sensitive to Bafilomycin A1, an inhibitor of VHA. We also report the first evidence of splice variants of the sodium/proton exchanger, NHE3, with both high and low molecular weight variants highly expressed on the apical membrane of the posterior rectal cells. Evidence of NHE3 function was indicated with in situ sodium transport significantly inhibited by a NHE3 antagonist, S3226. We propose that the outward proton pumping by VHA establishes a favourable electromotive gradient to drive sodium secretion via NHE3 thus producing a hyperosmotic, sodium-rich urine. This H+- driven Na+ secretion process is the primary mechanism of ion regulation in salt-tolerant culicine mosquito species and was first investigated over 80 years ago.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- Hill, R. W., Wyse, G. A. & Anderson, M. Animal Physiology 4th edn. (Sinauer Associates, Inc. Publishers, 2016).
-
- Nation, J. L. Insect Physiology and Biochemistry 3rd edn. (CRC Press, Taylor & Francis Group, 2016).
-
- Stobbart, R. H. & Shaw, J. in The Physiology of Insecta, Vol. 3, 190–255 (ed Rockstein, M.) Ch. 4 (Academic Press, 1964).
-
- Beadle, L. C. Regulation of the haemolymph in the saline water mosquito larvae Aedes detritus Edw. J. Exp. Biol.16, 346–362 (1939). - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

