Multi-arm multi-stage (MAMS) randomised selection designs: impact of treatment selection rules on the operating characteristics
- PMID: 38831421
- PMCID: PMC11145876
- DOI: 10.1186/s12874-024-02247-w
Multi-arm multi-stage (MAMS) randomised selection designs: impact of treatment selection rules on the operating characteristics
Erratum in
-
Correction: Multi‑arm multi‑stage (MAMS) randomised selection designs: impact of treatment selection rules on the operating characteristics.BMC Med Res Methodol. 2024 Jul 2;24(1):143. doi: 10.1186/s12874-024-02267-6. BMC Med Res Methodol. 2024. PMID: 38956455 Free PMC article. No abstract available.
Abstract
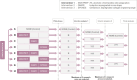
Background: Multi-arm multi-stage (MAMS) randomised trial designs have been proposed to evaluate multiple research questions in the confirmatory setting. In designs with several interventions, such as the 8-arm 3-stage ROSSINI-2 trial for preventing surgical wound infection, there are likely to be strict limits on the number of individuals that can be recruited or the funds available to support the protocol. These limitations may mean that not all research treatments can continue to accrue the required sample size for the definitive analysis of the primary outcome measure at the final stage. In these cases, an additional treatment selection rule can be applied at the early stages of the trial to restrict the maximum number of research arms that can progress to the subsequent stage(s). This article provides guidelines on how to implement treatment selection within the MAMS framework. It explores the impact of treatment selection rules, interim lack-of-benefit stopping boundaries and the timing of treatment selection on the operating characteristics of the MAMS selection design.
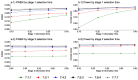
Methods: We outline the steps to design a MAMS selection trial. Extensive simulation studies are used to explore the maximum/expected sample sizes, familywise type I error rate (FWER), and overall power of the design under both binding and non-binding interim stopping boundaries for lack-of-benefit.
Results: Pre-specification of a treatment selection rule reduces the maximum sample size by approximately 25% in our simulations. The familywise type I error rate of a MAMS selection design is smaller than that of the standard MAMS design with similar design specifications without the additional treatment selection rule. In designs with strict selection rules - for example, when only one research arm is selected from 7 arms - the final stage significance levels can be relaxed for the primary analyses to ensure that the overall type I error for the trial is not underspent. When conducting treatment selection from several treatment arms, it is important to select a large enough subset of research arms (that is, more than one research arm) at early stages to maintain the overall power at the pre-specified level.
Conclusions: Multi-arm multi-stage selection designs gain efficiency over the standard MAMS design by reducing the overall sample size. Diligent pre-specification of the treatment selection rule, final stage significance level and interim stopping boundaries for lack-of-benefit are key to controlling the operating characteristics of a MAMS selection design. We provide guidance on these design features to ensure control of the operating characteristics.
Keywords: Adaptive trial designs; Familywise type I error rate; MAMS; Multi-arm multi-stage randomised clinical trials; ROSSINI-2 trial; Treatment selection.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Choodari-Oskooei B, Sydes M, Royston P, Parmar MKB. Principles and Practice of Clinical Trials. 1. Cham: Springer; 2022. Multi-arm Multi-stage (MAMS) Platform Randomized Clinical Trials.
-
- ROSSINI 2 - Reduction of Surgical Site Infection Using Several Novel Interventions. https://clinicaltrials.gov/ct2/show/NCT03838575. Accessed 17 June 2019.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

