Gasdermin D deficiency aborts myeloid calcium influx to drive granulopoiesis in lupus nephritis
- PMID: 38831451
- PMCID: PMC11149269
- DOI: 10.1186/s12964-024-01681-z
Gasdermin D deficiency aborts myeloid calcium influx to drive granulopoiesis in lupus nephritis
Abstract
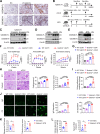
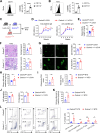
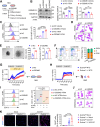
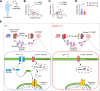
Gasdermin D (GSDMD) is emerging as an important player in autoimmune diseases, but its exact role in lupus nephritis (LN) remains controversial. Here, we identified markedly elevated GSDMD in human and mouse LN kidneys, predominantly in CD11b+ myeloid cells. Global or myeloid-conditional deletion of GSDMD was shown to exacerbate systemic autoimmunity and renal injury in lupus mice with both chronic graft-versus-host (cGVH) disease and nephrotoxic serum (NTS) nephritis. Interestingly, RNA sequencing and flow cytometry revealed that myeloid GSDMD deficiency enhanced granulopoiesis at the hematopoietic sites in LN mice, exhibiting remarkable enrichment of neutrophil-related genes, significant increases in total and immature neutrophils as well as granulocyte/macrophage progenitors (GMPs). GSDMD-deficient GMPs and all-trans-retinoic acid (ATRA)-stimulated human promyelocytes NB4 were further demonstrated to possess enhanced clonogenic and differentiation abilities compared with controls. Mechanistically, GSDMD knockdown promoted self-renewal and granulocyte differentiation by restricting calcium influx, contributing to granulopoiesis. Functionally, GSDMD deficiency led to increased pathogenic neutrophil extracellular traps (NETs) in lupus peripheral blood and bone marrow-derived neutrophils. Taken together, our data establish that GSDMD deletion accelerates LN development by promoting granulopoiesis in a calcium influx-regulated manner, unraveling its unrecognized critical role in LN pathogenesis.
Keywords: Calcium influx; Gasdermin D; Granulopoiesis; Lupus nephritis; Myeloid cell.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Deletion of gasdermin D promotes granulocytic myeloid-derived suppressor cell differentiation by decreased release of mitochondrial DNA to promote tumor escape.Cancer Immunol Immunother. 2025 Jul 25;74(9):277. doi: 10.1007/s00262-025-04104-1. Cancer Immunol Immunother. 2025. PMID: 40711527 Free PMC article.
-
STING regulates NETs formation by activating GSDMD in influenza viral pneumonia.Front Immunol. 2025 Jul 1;16:1598902. doi: 10.3389/fimmu.2025.1598902. eCollection 2025. Front Immunol. 2025. PMID: 40666504 Free PMC article.
-
Gasdermin D silencing alleviates airway inflammation and remodeling in an ovalbumin-induced asthmatic mouse model.Cell Death Dis. 2024 Jun 7;15(6):400. doi: 10.1038/s41419-024-06777-5. Cell Death Dis. 2024. PMID: 38849380 Free PMC article.
-
Review: the role of GSDMD in sepsis.Inflamm Res. 2022 Nov;71(10-11):1191-1202. doi: 10.1007/s00011-022-01624-9. Epub 2022 Aug 15. Inflamm Res. 2022. PMID: 35969260 Free PMC article.
-
FCGR3B copy number loss rather than gain is a risk factor for systemic lupus erythematous and lupus nephritis: a meta-analysis.Int J Rheum Dis. 2015 May;18(4):392-7. doi: 10.1111/1756-185X.12342. Epub 2014 Mar 27. Int J Rheum Dis. 2015. PMID: 24673810
Cited by
-
New Mechanisms and Therapeutic Targets in Systemic Lupus Erythematosus.MedComm (2020). 2025 Jun 9;6(6):e70246. doi: 10.1002/mco2.70246. eCollection 2025 Jun. MedComm (2020). 2025. PMID: 40491969 Free PMC article. Review.
-
Innate immune cells in acute and chronic kidney disease.Nat Rev Nephrol. 2025 Jul;21(7):464-482. doi: 10.1038/s41581-025-00958-x. Epub 2025 Apr 22. Nat Rev Nephrol. 2025. PMID: 40263532 Review.
References
-
- Kiriakidou M, Ching CL: Systemic Lupus Erythematosus. Ann Intern Med 2020, 172(11):ITC81-ITC96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 2020B1212060028/NHC Key Laboratory of Clinical Nephrology (Sun Yat-Sen University) and Guangdong Provincial Key Laboratory of Nephrology
- 82270716/National Natural Science Foundation of China
- 82270764/National Natural Science Foundation of China
- 2017A030306013/Natural Science Foundation of Guangdong Province
- 2017TQ04R549/Guangdong Special Support Program
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

