Prefrontal cortical ripples mediate top-down suppression of hippocampal reactivation during sleep memory consolidation
- PMID: 38834064
- PMCID: PMC11233241
- DOI: 10.1016/j.cub.2024.05.018
Prefrontal cortical ripples mediate top-down suppression of hippocampal reactivation during sleep memory consolidation
Abstract
Consolidation of initially encoded hippocampal representations in the neocortex through reactivation is crucial for long-term memory formation and is facilitated by the coordination of hippocampal sharp-wave ripples (SWRs) with cortical slow and spindle oscillations during non-REM sleep. Recent evidence suggests that high-frequency cortical ripples can also coordinate with hippocampal SWRs in support of consolidation; however, the contribution of cortical ripples to reactivation remains unclear. We used high-density, continuous recordings in the hippocampus (area CA1) and prefrontal cortex (PFC) over the course of spatial learning and show that independent PFC ripples dissociated from SWRs are prevalent in NREM sleep and predominantly suppress hippocampal activity. PFC ripples paradoxically mediate top-down suppression of hippocampal reactivation rather than coordination, and this suppression is stronger for assemblies that are reactivated during coordinated CA1-PFC ripples for consolidation of recent experiences. Further, we show non-canonical, serial coordination of independent cortical ripples with slow and spindle oscillations, which are known signatures of memory consolidation. These results establish a role for prefrontal cortical ripples in top-down regulation of behaviorally relevant hippocampal representations during consolidation.
Keywords: consolidation; hippocampus; non-REM; prefrontal cortex; reactivation; replay; ripples; sleep; suppression; top-down.
Copyright © 2024 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures
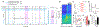
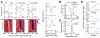
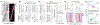

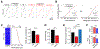
Update of
-
Cortical ripples mediate top-down suppression of hippocampal reactivation during sleep memory consolidation.bioRxiv [Preprint]. 2023 Dec 13:2023.12.12.571373. doi: 10.1101/2023.12.12.571373. bioRxiv. 2023. Update in: Curr Biol. 2024 Jul 8;34(13):2801-2811.e9. doi: 10.1016/j.cub.2024.05.018. PMID: 38168420 Free PMC article. Updated. Preprint.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

