Neuropeptide Modulation Enables Biphasic Internetwork Coordination via a Dual-Network Neuron
- PMID: 38834302
- PMCID: PMC11211724
- DOI: 10.1523/ENEURO.0121-24.2024
Neuropeptide Modulation Enables Biphasic Internetwork Coordination via a Dual-Network Neuron
Abstract
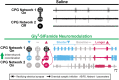
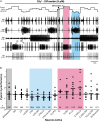
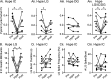
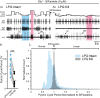
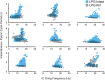
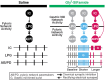
Linked rhythmic behaviors, such as respiration/locomotion or swallowing/chewing, often require coordination for proper function. Despite its prevalence, the cellular mechanisms controlling coordination of the underlying neural networks remain undetermined in most systems. We use the stomatogastric nervous system of the crab Cancer borealis to investigate mechanisms of internetwork coordination, due to its small, well-characterized feeding-related networks (gastric mill [chewing, ∼0.1 Hz]; pyloric [filtering food, ∼1 Hz]). Here, we investigate coordination between these networks during the Gly1-SIFamide neuropeptide modulatory state. Gly1-SIFamide activates a unique triphasic gastric mill rhythm in which the typically pyloric-only LPG neuron generates dual pyloric-plus gastric mill-timed oscillations. Additionally, the pyloric rhythm exhibits shorter cycles during gastric mill rhythm-timed LPG bursts, and longer cycles during IC, or IC plus LG gastric mill neuron bursts. Photoinactivation revealed that LPG is necessary to shorten pyloric cycle period, likely through its rectified electrical coupling to pyloric pacemaker neurons. Hyperpolarizing current injections demonstrated that although LG bursting enables IC bursts, only gastric mill rhythm bursts in IC are necessary to prolong the pyloric cycle period. Surprisingly, LPG photoinactivation also eliminated prolonged pyloric cycles, without changing IC firing frequency or gastric mill burst duration, suggesting that pyloric cycles are prolonged via IC synaptic inhibition of LPG, which indirectly slows the pyloric pacemakers via electrical coupling. Thus, the same dual-network neuron directly conveys excitation from its endogenous bursting and indirectly funnels synaptic inhibition to enable one network to alternately decrease and increase the cycle period of a related network.
Keywords: central pattern generator; internetwork; neuromodulation; neuropeptide; rectification; rhythmic.
Copyright © 2024 Gnanabharathi et al.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



Similar articles
-
Neuropeptide modulation of bidirectional internetwork synapses.J Neurophysiol. 2024 Jul 1;132(1):184-205. doi: 10.1152/jn.00149.2024. Epub 2024 May 22. J Neurophysiol. 2024. PMID: 38776457
-
Neuronal Switching between Single- and Dual-Network Activity via Modulation of Intrinsic Membrane Properties.J Neurosci. 2021 Sep 15;41(37):7848-7863. doi: 10.1523/JNEUROSCI.0286-21.2021. Epub 2021 Aug 4. J Neurosci. 2021. PMID: 34349000 Free PMC article.
-
Switching neuron contributions to second network activity.J Neurophysiol. 2024 Feb 1;131(2):417-434. doi: 10.1152/jn.00373.2023. Epub 2024 Jan 10. J Neurophysiol. 2024. PMID: 38197163 Free PMC article.
-
Frequency control of a slow oscillatory network by a fast rhythmic input: pyloric to gastric mill interactions in the crab stomatogastric nervous system.Ann N Y Acad Sci. 1998 Nov 16;860:226-38. doi: 10.1111/j.1749-6632.1998.tb09052.x. Ann N Y Acad Sci. 1998. PMID: 9928315 Review.
-
Modulation of stomatogastric rhythms.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2009 Nov;195(11):989-1009. doi: 10.1007/s00359-009-0483-y. Epub 2009 Oct 11. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2009. PMID: 19823843 Review.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
