Invasive pneumococcal disease surveillance in Canada, 2021-2022
- PMID: 38835503
- PMCID: PMC11147492
- DOI: 10.14745/ccdr.v50i05a02
Invasive pneumococcal disease surveillance in Canada, 2021-2022
Abstract
Background: Invasive pneumococcal disease (IPD, Streptococcus pneumoniae) has been a nationally notifiable disease in Canada since 2000. The use of conjugate vaccines has caused a shift in the distribution of serotypes over time. This report is a summary of the demographics, serotypes and antimicrobial resistance of IPD isolates collected in Canada in 2021 and 2022.
Methods: The National Microbiology Laboratory (NML) of the Public Health Agency of Canada in Winnipeg, Manitoba collaborates with provincial and territorial public health laboratories to conduct national surveillance of IPD. There were 1,999 isolates reported in 2021 and 3,775 isolates in 2022. Serotype was determined by the Quellung reaction or whole-genome sequencing (WGS). Antimicrobial susceptibilities were determined by WGS methods, broth microdilution, or data shared by collaborators in the Canadian Antimicrobial Resistance Alliance program at the University of Manitoba. Population-based IPD incidence rates were obtained through the Canadian Notifiable Disease Surveillance System.
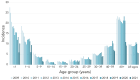
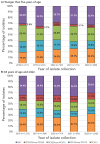
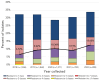
Results: The incidence of IPD in Canada was 5.62 cases per 100,000 population in 2021, decreasing from the peak of 10.86 cases per 100,000 population in 2018. Serotypes with increasing trends (p<0.05) between 2018 and 2022 included: 4 (6.1%-12.4%), 9V (1.0%-5.1%) and 12F (4.8%-5.4%). The overall prevalence of PCV13 serotypes increased over the same period (31.2%-41.5%, p<0.05) while the prevalence of non-vaccine types decreased significantly (27.3%-21.5%, p<0.0001). The highest rates of antimicrobial resistance in 2021 and 2022 were seen with clarithromycin (21%, 2021; 24%, 2022) and erythromycin (22%, 2021; 24%, 2022). Multidrug-resistant IPD continued to increase from 2018 to 2022 (6.7%-12.6%, p<0.05).
Conclusion: The number of cases of IPD continued to decrease in 2021 in comparison to previous years, however, 2022 saw a return to pre-COVID-19 levels. Disease due to PCV13 serotypes 3, 4, 9V and 19F, as well as non-PCV13 serotypes 12F and 20, is increasing in prevalence. Surveillance of IPD to monitor changing serotype distribution and antimicrobial resistance is essential.
Keywords: Canada; IPD; PCV13; Streptococcus pneumoniae; antimicrobial resistance; invasive pneumococcal disease; pneumococcus; serotype; surveillance.
Conflict of interest statement
Competing interests None.
Figures

References
-
- Ganaie F, Saad JS, McGee L, van Tonder AJ, Bentley SD, Lo SW, Gladstone RA, Turner P, Keenan JD, Breiman RF, Nahm MH. A New Pneumococcal Capsule Type, 10D, is the 100th Serotype and Has a Large cps Fragment from an Oral Streptococcus. MBio 2020;11(3):e00937–20. 10.1128/mBio.00937-20 - DOI - PMC - PubMed
-
- Bettinger JA, Scheifele DW, Kellner JD, Halperin SA, Vaudry W, Law B, Tyrrell G; Canadian Immunization Monitoring Program, Active (IMPACT) . The effect of routine vaccination on invasive pneumococcal infections in Canadian children, Immunization Monitoring Program, Active 2000-2007. Vaccine 2010;28(9):2130–6. 10.1016/j.vaccine.2009.12.026 - DOI - PubMed
-
- Demczuk WH, Martin I, Griffith A, Lefebvre B, McGeer A, Lovgren M, Tyrrell GJ, Desai S, Sherrard L, Adam H, Gilmour M, Zhanel GG; Toronto Bacterial Diseases Network; Canadian Public Health Laboratory Network . Serotype distribution of invasive Streptococcus pneumoniae in Canada after the introduction of the 13-valent pneumococcal conjugate vaccine, 2010-2012. Can J Microbiol 2013;59(12):778–88. 10.1139/cjm-2013-0614 - DOI - PubMed
-
- Desai S, McGeer A, Quach-Thanh C, Elliott D. Update on the Use of Conjugate Pneumococcal Vaccines in Childhood: An Advisory Committee Statement (ACS). National Advisory Committee on Immunization (NACI). Can Commun Dis Rep 2010;36(ACS-12):1–21. 10.14745/ccdr.v36i00a1210.14745/ccdr.v36i00a12 - DOI - DOI - PMC - PubMed