Non-invasive prenatal testing: when results suggests maternal cancer
- PMID: 38835737
- PMCID: PMC11006267
- DOI: 10.1515/medgen-2023-2055
Non-invasive prenatal testing: when results suggests maternal cancer
Abstract
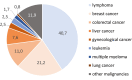
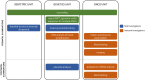
It is now well-established that non-invasive prenatal testing (NIPT), originally designed to screen cell-free DNA (cfDNA) in maternal blood for the presence of common fetal trisomies, can lead to incidental detection of occult maternal malignancies. Retrospective evaluations have demonstrated that the detection of multiple copy number alterations in cfDNA is particularly suggestive of an incipient tumor and that cancer detection rates not only depend on tumor biology but also on applied NIPT technologies and downstream diagnostic investigations. Since the identification of a maternal cancer in pregnancy has implications for both woman and the unborn child, prospective studies are needed to provide evidence on best clinical practices and on clinical utility in terms of patient outcomes.
Keywords: Cell-free DNA; incidental finding; maternal malignancy; non-invasive prenatal testing.
© 2023 bei den Autoren, publiziert von De Gruyter.
Conflict of interest statement
Competing interests: The authors declare no conflict of interest.
Figures





Similar articles
-
Incidental Diagnosis of Occult Maternal Malignancy With Routine Noninvasive Prenatal Testing During Pregnancy.Cureus. 2024 Dec 24;16(12):e76331. doi: 10.7759/cureus.76331. eCollection 2024 Dec. Cureus. 2024. PMID: 39850155 Free PMC article.
-
Noninvasive Prenatal Testing and Incidental Detection of Occult Maternal Malignancies.JAMA. 2015 Jul 14;314(2):162-9. doi: 10.1001/jama.2015.7120. JAMA. 2015. PMID: 26168314
-
Non-invasive prenatal testing suggesting a maternal malignancy: What do we tell the prospective parents in Belgium?Prenat Diagn. 2021 Sep;41(10):1264-1272. doi: 10.1002/pd.6031. Epub 2021 Aug 26. Prenat Diagn. 2021. PMID: 34405430 Review.
-
Genome-wide non-invasive prenatal testing in single- and multiple-pregnancies at any risk: Identification of maternal polymorphisms to reduce the number of unnecessary invasive confirmation testing.Eur J Obstet Gynecol Reprod Biol. 2020 Sep;252:19-29. doi: 10.1016/j.ejogrb.2020.05.070. Epub 2020 Jun 2. Eur J Obstet Gynecol Reprod Biol. 2020. PMID: 32619881
-
Maternal Plasma DNA and RNA Sequencing for Prenatal Testing.Adv Clin Chem. 2016;74:63-102. doi: 10.1016/bs.acc.2015.12.004. Epub 2016 Jan 21. Adv Clin Chem. 2016. PMID: 27117661 Review.
Cited by
-
Liquid Biopsy: minimal-invasive analysis of somatic alterations.Med Genet. 2023 Dec 5;35(4):199-200. doi: 10.1515/medgen-2023-2061. eCollection 2023 Dec. Med Genet. 2023. PMID: 38835738 Free PMC article. No abstract available.
-
Non-Invasive Prenatal Screening from a Genetic Counseling Prospective: Pre and Post-Genetic Counseling Regarding Rare Chromosomal Abnormalities and Incidental Finding.Genes (Basel). 2024 Oct 21;15(10):1349. doi: 10.3390/genes15101349. Genes (Basel). 2024. PMID: 39457473 Free PMC article. Review.
References
-
- Dennis Lo Y. M. ‘Presence of fetal DNA in maternal plasma and serum’. Lancet, vol. 350, no. 9076. Aug. 1997. pp. 485–487. et al. doi: 10.1016/S0140-6736(97)02174-0. - PubMed
-
- Brison N. ‘Accuracy and clinical value of maternal incidental findings during noninvasive prenatal testing for fetal aneuploidies’. Genet Med, vol. 19, no. 3. Mar. 2017. pp. 306–313. et al. doi: 10.1038/GIM.2016.113. - PubMed
LinkOut - more resources
Full Text Sources
