Multi-Center Two-Year Patency Outcomes of Endovascular Arteriovenous Fistulas (endoAVF) Created with a 4 French System
- PMID: 38839640
- PMCID: PMC11303495
- DOI: 10.1007/s00270-024-03754-5
Multi-Center Two-Year Patency Outcomes of Endovascular Arteriovenous Fistulas (endoAVF) Created with a 4 French System
Abstract
Purpose: To assess multicenter two-year patency outcomes of endovascular arteriovenous fistulas (endoAVF) created with the WavelinQ device.
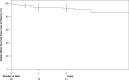
Materials and methods: Patients who had fistulas created at three centers from January 2018 to December 2020 were included in this retrospective study. In total, 112 patients underwent endoAVF creation [40 females, 72 males; mean age 60 years (range 18-88)]. Data collected included patient demographics, location of fistula creation, interventions performed, and brachial artery flows pre- and post-creation. Two-year cumulative patency, functional patency, and primary patency were assessed with Kaplan-Meier methodology. Factors affecting patency and maturation were examined using the Cox proportional hazards model.
Results: Technical success defined as angiographically successful endoAVF creation was 97.3% (109/112). In 11 patients the fistula did not mature for dialysis use. For 98 patients (87%) with endoAVF maturation, 12- and 24-month cumulative patency was 94.3% and 91.7%. Functional patency (two-needle cannulation) at 12 and 24 months was 95.7% and 92.7%, respectively. Median maturation time is 95 days (IQR 51-231 days). Male gender and brachial vein coiling at the time of endoAVF creation were predictive of maturation. There were 34 censored events (four patients undergoing renal transplantation; 30 patients deceased). Number of reinterventions per patient year was 0.73 where 43 were maturation procedures and 101 were maintenance procedures. One Grade 3 complication occurred of arterial access puncture site pseudoaneurysm.
Conclusion: A high two-year functional and cumulative patency following endoAVF creation with the WavelinQ device was observed in this multicenter real-world experience Level of Evidence: 3 Level of Evidence III.
Keywords: Arteriovenous fistula; Endoavf; Hemodialysis; Percutaneous; Wavelinq.
© 2024. The Author(s).
Conflict of interest statement
All authors are consultants for BD except EK.
Figures


References
-
- Pecoits-Filho R, Okpechi IG, Donner JA, et al. Capturing and monitoring global differences in untreated and treated end-stage kidney disease, kidney replacement therapy modality, and outcomes. Kidney Int Suppl. 2020;10(1):e3–9. 10.1016/j.kisu.2019.11.001.10.1016/j.kisu.2019.11.001 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

