Field study to determine the reliability of HIV viral load results shows minimal impact of delayed testing in South Africa
- PMID: 38840959
- PMCID: PMC11151376
- DOI: 10.4102/ajlm.v13i1.2364
Field study to determine the reliability of HIV viral load results shows minimal impact of delayed testing in South Africa
Abstract
Background: Understanding factors that impact HIV viral load (VL) accuracy in resource-limited settings is key to quality improvement.
Objective: We evaluated whether testing delay and specimen storage between 25 °C and 30 °C before testing affected results.
Methods: Between November 2019 and June 2023, 249 individuals on antiretroviral therapy, or with newly diagnosed HIV, were recruited from clinics in Cape Town and Gqeberha, South Africa, and three plasma preparation tubes were collected. One tube was tested within 24 h, while the others were stored uncentrifuged at ambient temperatures before testing. Centrifugation and testing of matched samples were performed on Day 4 and Day 7 after collection.
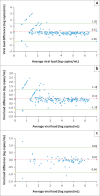
Results: Time delay and ambient storage had minimal impact in specimens with a Day 1 VL of > 100 copies/mL. When grouped by Day 1 VL range, 96% - 100% of specimens at Day 4 and 93% - 100% at Day 7 had VLs within 0.5 log copies/mL of the first result. The greatest variability at Days 4 and 7 was observed when the Day 1 VL was < 100 copies/mL. However, there was no trend of increasing difference over time. Of Day 1 specimens with undetectable VL, or VL < 50 copies/mL, 80% had concordant results at Day 4 and 78% at Day 7.
Conclusion: These results show that VL is stable in plasma preparation tubes for 7 days when stored at room temperature. There is significant variability in specimens with low VL, but variability is not affected by testing delay.
What this study adds: Ideal HIV VL testing conditions are frequently unachievable in resource-limited settings. Data are needed on whether this impacts on the validity of test results. Our results provide reassurance that storage at ambient temperature for up to 7 days before testing does not substantially affect the VL result.
Keywords: HIV viral load stability; delayed testing; diagnostic accuracy; high-throughput viral load testing; plasma preparation tubes.
© 2024. The Authors.
Conflict of interest statement
The authors declare that they have no financial or personal relationships that may have inappropriately influenced them in writing this article.
Figures

References
-
- Robert B, Brown EB. FAST-TRACK ending the AIDS epidemic by 2030. UNAIDS Publ [serial online]. 2014;(1):1–14. [cited 2024 Apr 01]. Available from: https://www.unaids.org/sites/default/files/media_asset/JC2686_WAD2014rep...
-
- NHLS Annual report 2017/18 [homepage on the Internet]. 2018, p. 71–75. [cited 2024 Apr 01]. Available from: http://intranet.nhls.ac.za/assets/files/policy/NHLS_AR_2018.pdf
-
- Abbott RealTime HIV1 kit insert 51-602100/R10 [homepage on the Internet]. 2014. [cited 2024 Apr 01]. Available from: https://www.molecular.abbott/content/dam/add/molecular/products/pdf-/rea...
-
- Roche CAP/CTM HIV-1 v2.0 EXPT-IVD [homepage on the Internet]. 2018. [cited 2024 Apr 01]. Available from: https://elabdoc-prod.roche.com/eLD/api/downloads/ee10c6ed-0bff-e911-fa90...
LinkOut - more resources
Full Text Sources
