Xylitol is prothrombotic and associated with cardiovascular risk
- PMID: 38842092
- PMCID: PMC11492277
- DOI: 10.1093/eurheartj/ehae244
Xylitol is prothrombotic and associated with cardiovascular risk
Abstract
Background and aims: The pathways and metabolites that contribute to residual cardiovascular disease risks are unclear. Low-calorie sweeteners are widely used sugar substitutes in processed foods with presumed health benefits. Many low-calorie sweeteners are sugar alcohols that also are produced endogenously, albeit at levels over 1000-fold lower than observed following consumption as a sugar substitute.
Methods: Untargeted metabolomics studies were performed on overnight fasting plasma samples in a discovery cohort (n = 1157) of sequential stable subjects undergoing elective diagnostic cardiac evaluations; subsequent stable isotope dilution liquid chromatography tandem mass spectrometry (LC-MS/MS) analyses were performed on an independent, non-overlapping validation cohort (n = 2149). Complementary isolated human platelet, platelet-rich plasma, whole blood, and animal model studies examined the effect of xylitol on platelet responsiveness and thrombus formation in vivo. Finally, an intervention study was performed to assess the effects of xylitol consumption on platelet function in healthy volunteers (n = 10).
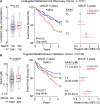
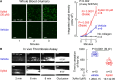
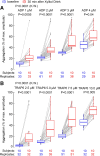
Results: In initial untargeted metabolomics studies (discovery cohort), circulating levels of a polyol tentatively assigned as xylitol were associated with incident (3-year) major adverse cardiovascular event (MACE) risk. Subsequent stable isotope dilution LC-MS/MS analyses (validation cohort) specific for xylitol (and not its structural isomers) confirmed its association with incident MACE risk [third vs. first tertile adjusted hazard ratio (95% confidence interval), 1.57 (1.12-2.21), P < .01]. Complementary mechanistic studies showed xylitol-enhanced multiple indices of platelet reactivity and in vivo thrombosis formation at levels observed in fasting plasma. In interventional studies, consumption of a xylitol-sweetened drink markedly raised plasma levels and enhanced multiple functional measures of platelet responsiveness in all subjects.
Conclusions: Xylitol is associated with incident MACE risk. Moreover, xylitol both enhanced platelet reactivity and thrombosis potential in vivo. Further studies examining the cardiovascular safety of xylitol are warranted.
Keywords: Artificial sweetener; Cardiovascular disease; Heart attack; Low-calorie sweetener; Nutrition; Platelet; Polyol; Stroke; Sugar alcohol; Thrombosis.
© The Author(s) 2024. Published by Oxford University Press on behalf of the European Society of Cardiology. All rights reserved. For commercial re-use, please contact reprints@oup.com for reprints and translation rights for reprints. All other permissions can be obtained through our RightsLink service via the Permissions link on the article page on our site—for further information please contact journals.permissions@oup.com.
Figures




References
-
- Abarca-Gómez L, Abdeen ZA, Hamid ZA, Abu-Rmeileh NM, Acosta-Cazares B, Acuin C, et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet 2017;390:2627–42. 10.1016/S0140-6736(17)32129-3 - DOI - PMC - PubMed
-
- Mortensen A. Sweeteners permitted in the European Union: safety aspects. Scand J Food Nutr 2006;50:104–16. 10.1080/17482970600982719 - DOI
-
- World Health Organization . Use of Non-Sugar Sweeteners: WHO Guideline: World Health Organization, 2023. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

