Nanoliposomal Irinotecan With Fluorouracil and Leucovorin or Gemcitabine Plus Cisplatin in Advanced Cholangiocarcinoma: A Phase II Study of the AIO Hepatobiliary-YMO Cancer Groups (NIFE-AIO-YMO HEP-0315)
- PMID: 38843469
- PMCID: PMC11379362
- DOI: 10.1200/JCO.23.01566
Nanoliposomal Irinotecan With Fluorouracil and Leucovorin or Gemcitabine Plus Cisplatin in Advanced Cholangiocarcinoma: A Phase II Study of the AIO Hepatobiliary-YMO Cancer Groups (NIFE-AIO-YMO HEP-0315)
Abstract
Purpose: First-line therapy options in advanced cholangiocarcinoma (CCA) are based on the ABC-02 trial regimen (gemcitabine/cisplatin [G/C]). The NIFE trial examined nanoliposomal irinotecan/fluorouracil/leucovorin (nal-IRI/FU/LV) as alternative first-line therapy in advanced CCA.
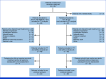
Methods: NIFE is a prospective, open-label, randomized, multicenter phase II study that aimed at detecting efficacy comparable with the standard treatment. Patients with advanced CCA were randomly assigned (1:1) to receive nal-IRI/FU/LV (arm A) or G/C (arm B). Stratification parameters were intrahepatic versus extrahepatic CCA, sex, and Eastern Cooperative Oncology Group (ECOG; 0/1). Arm A was designed as a Simon's optimal two-stage design and arm B served as a randomized control group. The primary goal was to exclude an inferior progression-free survival (PFS) at 4 months of only 40%, while assuming a rate of 60% on G/C population.
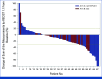
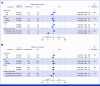
Results: Between 2018 and 2020, overall 91 patients were randomly assigned to receive nal-IRI/FU/LV (n = 49) or G/C (n = 42). The NIFE trial formally met its primary end point with a 4-month PFS rate of 51% in patients receiving nal-IRI/FU/LV. The median PFS was 6 months (2.4-9.6) in arm A and 6.9 months (2.5-7.9) in arm B. Median overall survival (OS) was 15.9 months (10.6-20.3) in arm A and 13.6 months (6.5-17.7) in arm B. The exploratory comparison of study arms suggested a numerical but statistically not significant advantage for nal-IRI/FU/LV (hazard ratio for PFS, 0.85 [95% CI, 0.53 to 1.38] and for OS, 0.94 [95% CI, 0.58 to 1.50]). Analysis for stratification parameters revealed no differences for sex and ECOG, but for tumor localization. The objective response rate was 24.5% with nal-IRI/FU/LV and 11.9% with G/C. No unexpected toxicities occurred. AEs related to nal-IRI/FU/LV were mainly GI and to G/C hematologic.
Conclusion: Treatment of advanced CCA with nal-IRI/FU/LV demonstrated efficacy in first-line therapy without new safety findings and merits further validation.
Trial registration: ClinicalTrials.gov NCT03044587.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
No other potential conflicts of interest were reported.
Figures


References
-
- Siegel RL, Miller KD, Jemal A: Cancer statistics, 2017. CA Cancer J Clin 67:7-30, 2017 - PubMed
-
- von Hahn T, Ciesek S, Wegener G, et al. : Epidemiological trends in incidence and mortality of hepatobiliary cancers in Germany. Scand J Gastroenterol 46:1092-1098, 2011 - PubMed
-
- Voesch S, Bitzer M, Blodt S, et al. : S3-Leitlinie: Diagnostik und Therapie des hepatozellulären Karzinoms und biliärer Karzinome–Version 2.0–Juni 2021, AWMF-Registernummer: 032-053OL. Z Gastroenterol 60:e131-e185, 2022 - PubMed
-
- Valle JW, Borbath I, Khan SA, et al. : Biliary cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 27:v28-v37, 2016. (suppl 5) - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

